Circular gap closure assay
Purse string-mediated wound healing
Purse string-mediated wound healing
Gap closure, actin purse-string contraction, wound healing.
In standard cultures, cells cannot be organized to mimic a gap within a tissue.
The closure of such a discontinuity can be relevant to model several biological processes occurring in vivo during embryogenesis, tissue morphogenesis or wound healing.
When cultured around circular non-adhesive substrates, cells can collectively contract using a supracellular actomyosin ring, to close non-adherent gaps.
Cells that are involved in wound healing, such as skin keratinocytes [1], epithelial kidney cells [2] or embryonic epidermal cells, are able to close non-adhesive gaps using actomyosin-mediated purse-string contraction. As the cells proliferate and form stable cell-cell connections, supracellular events are triggered, such as actin structures over the entire inner periphery of the non-adhesive area, that work to close the gap (Figure 1).
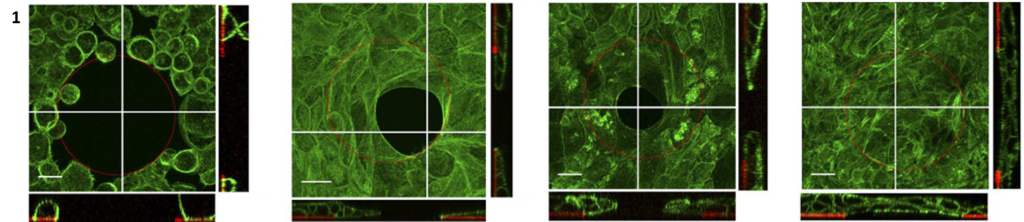
Keratinocytes closing a 100 µm diameter non-adhesive gap using actin purse-string contraction [1].
This phenomenon can be reproduced using 4Dcell micropatterns.
Mouse embryonic fibroblasts form well-defined circular assemblies of cells on ring-shaped micropatterns (inner diameter 300 um, see left panel of Figure 2). These cells collectively contract to progressively close the gap within the cell monolayer. This process relies entirely on cell cooperation and contraction to close this gap: the cells inside the ring cannot adhere and are not migrating. They form an epithelial sheet suspended over the gap. This is evidenced by the bright circle at the inner periphery of the closing ring, suggestive of a thick layer of unadhered cells (Figure 2).
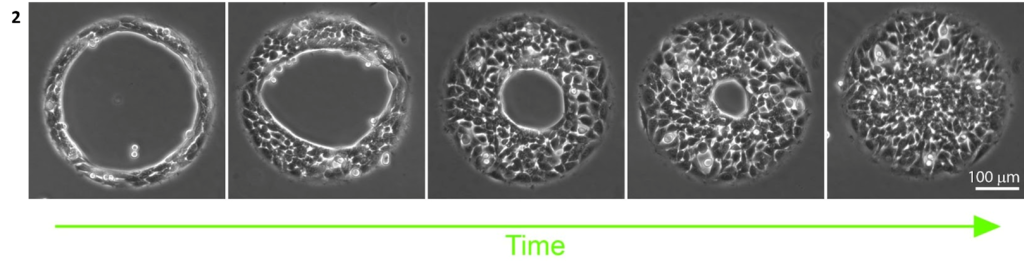
Mouse embryonic fibroblasts closing a 300 µm diameter non-adhesive gap [4Dcell data].