> FIBROBLASTS
Discovery of their properties and emphasis on the assays used to test them
> FIBROBLASTS
Discovery of their properties and emphasis on the assays used to test them
> ABSTRACT
Fibroblasts are mesenchymal cells, which provide support and structure to the functional cells of the organs. They are major producers of collagen and other extracellular matrix (ECM) components, and are able to shape the tissues. However, in the past years, it has become more and more clear that the fibroblasts not only ensure the collagen turnover, but that they also actively interact with other cells, and ensure tissue homeostasis. They are particularly important for the promotion and regulation of inflammation, and for the process of wound healing, which, if it fails, can progress to fibrosis. The study of fibroblasts is a currently topic, with assays ranging from classical 2D monolayer cell cultures to sophisticated 3D systems and co-cultures.
> INTRODUCTION
Fibroblasts are cells of mesenchymal origin, not terminally differentiated, and one of the most abundant types of cells in the connective tissues. They are large flat cells, which show elongations at the end of their bodies. Their nucleus is flat and oval (“Fibroblast”, Encyclopaedia Britannica).
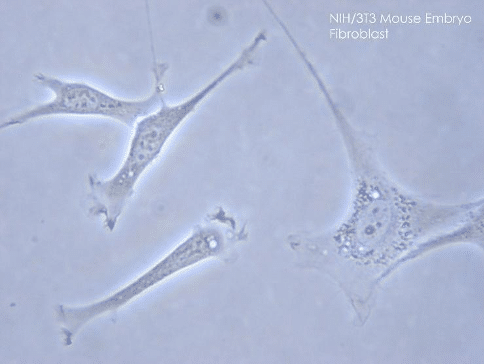
Figure 1. Phase contrast microscopy image of fibroblasts from a mouse embryo
Encyclopaedia britannica
They can be defined as the cells of the connective tissue which participate in its creation and maintenance, and which are not epithelial, vascular, or inflammatory cells (Kalluri & Zeisberg, 2006). Fibroblasts are found in most of the tissues of the human body. Their functions comprise especially deposition of ECM components, regulation of inflammation, regulation of epithelial differentiation, and are involved in wound healing. In other words, they ensure tissue homeostasis. However, if these processes are overactivated, they can lead, for instance, to tissue fibrosis, which is characterized by the formation of a permanent scar tissue.
There is high heterogeneity in the fibroblast populations, even within a single tissue. It has been shown that in the skin, for example, the fibroblast population arises from two lineages. One forms the upper dermis, and the other forms the lower dermis, and they have different functions. Indeed, the upper dermis is the centre for piloerection, and the fibroblasts of the lower dermis rather make the fibrillar ECM (Driskell et al., 2013).
Moreover, when talking about fibroblasts, we have to differentiate the resting fibroblasts, which ensure a sufficient turnover of ECM, and the activated fibroblasts, which differentiate into myofibroblasts (phenotypically between fibroblasts and smooth muscle cells). These cells express the α-smooth muscle actin (α-SMA) and can exert contractile forces, in addition to the production of ECM components. They are especially known for their role in normal and pathological wound healing, and are also present in a certain number of tissues in which they help them fulfil their functions (intestinal villous core for example) (Tomasek et al., 2002).
In this review, we are going to highlight some of the most characteristic physical abilities of the fibroblasts, and how these features play a role in the different processes in which they are involved in the body. We will have a particular focus on the type of assays used to test each of the physical characteristics, and how 4Dcell could reveal itself to be a particularly relevant actor in this research.
> PHYSICAL ABILITIES OF THE FIBROBLASTS
In this first part, we will go through the principal physical characteristics of the fibroblasts, which all together make them different from other types of cells. Mainly, we will talk about their capacity to create and propagate contractile forces to their surrounding environment, their ability to signal and to respond to chemical signals, their ability to propagate an electrical signal, and, last but not least, their capacity to synthesize collagen and other ECM components. We will discuss in the next part how these features play a role in the different processes in which the fibroblasts are involved in the body.
> Contractility
Back in 1980, Harris et al. showed that the pulling forces of cells, especially of fibroblasts, could distort a silicon rubber used as substrate (Harris et al., 1980). Later on, it was further confirmed that, rather than the shortening or dehydration of the material, it is the forces exerted by the cells which make the substrate shorten (Stopak & Harris, 1982). To prove it, researchers used collagen gels with randomly arranged fibres in which they were embedded. What they observed was a rearrangement of the fibres, due to the traction force exerted by the cells. In this first type of assays, the contraction is measured according to deformation and dimensional changes of the substrate. This first type of assays measures a relative difference in contraction according to different experimental conditions. A second type of experimental setup measures the contractile forces exerted by the cells, quantitatively this time. For example, Kolodney et al. used a silicone elastomer surrounding a collagen well in a petri dish, in contact with holders (see fig.2) (Kolodney & Wysolmerski, 1992).
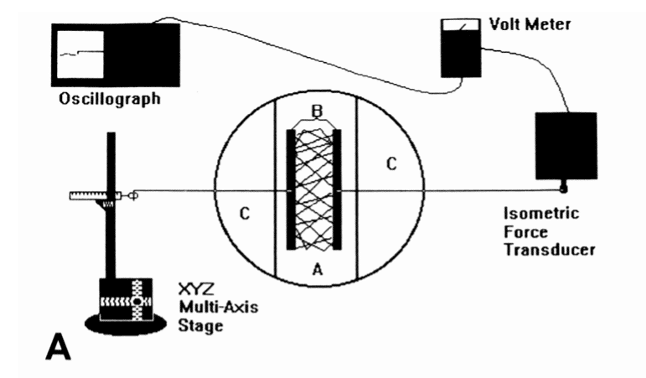
Figure 2. A schematic representation of the isometric force monitoring apparatus
Kolodney et al. (1992)
Thus, we can separate the assays between qualitative and quantitative ones. Another possible separation is between bulk and single cell measurements. The techniques we saw above are rather used for cellular bulk analysis, whereas micromachined cantilever beam array, micropost force sensor array (MFSA), cell traction force microscopy (CTFM), among others, can be used to perform single cell measurements (Li & Wang, 2011).
The traction forces created by the cells are thought to come, among other things, from the intracellular tension which is transmitted to the ECM via focal adhesions present at the end of stress fibres (Balaban et al., 2001). Actin polymerization, leading to cell migration, is also thought to be responsible for some forces applied to the ECM (Bereiter-Hahn, 2005). In addition, proteins like α-SMA, expressed in myofibroblasts, are also responsible for the generation of forces in the tissues (Hinz, Celetta, et al., 2001). Li and Wang (2009) found that the forces exerted by the fibroblasts embedded in a collagen lattice range from 11 to 42 nN , i.e., they are of the order of magnitude of 10-8 N (Li & Wang, 2011). As a comparison, the weight exerted by a cell is of the order of magnitude of 0.1 nN (for the calculation, the fibroblast has been approximated to a ball of water with a diameter of 20µm).
As contraction is an important feature of the fibroblasts, it is of interest to investigate the mechanisms of regulation of contraction. Grinnell et. al (1999) studied the contraction of the fibroblasts under different environmental conditions (Grinnell et al., 1999). Their results suggested that the fibroblasts regulate their contraction in response to different molecules, lysophosphatidic acid (LPA) and platelet-derived growth factor (PDGF) in the study, but with different kinetics whether they are embedded in floating or stressed collagen matrices. It highlights the fact that the fibroblasts not only generate forces, but that they are also able to sense the tensions from the environment, and that their mechanisms of regulation of their own contraction differ accordingly.
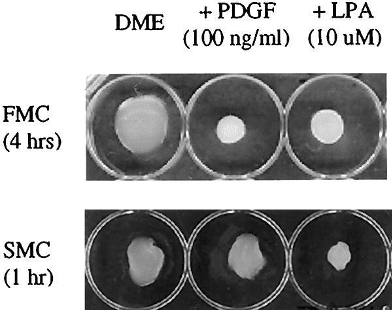
Figure 3. Appearance of floating (FMC) and stressed (SMC) matrices after contraction
Grinnell et al. (1999)
This ability to sense the mechanical forces is also brought out by the fact that it influences fibroblast differentiation into myofibroblasts (Hinz, Mastrangelo, et al., 2001). In their study, Hinz et al. showed that the mechanical tension provided by the environment is crucial for myofibroblast differentiation as well as for their contractility abilities. This was shown using an in vivo rat model, as opposed to the example mentioned above, in which α-SMA levels were measured in the cells from a wound, either relaxed or with mechanical tension.
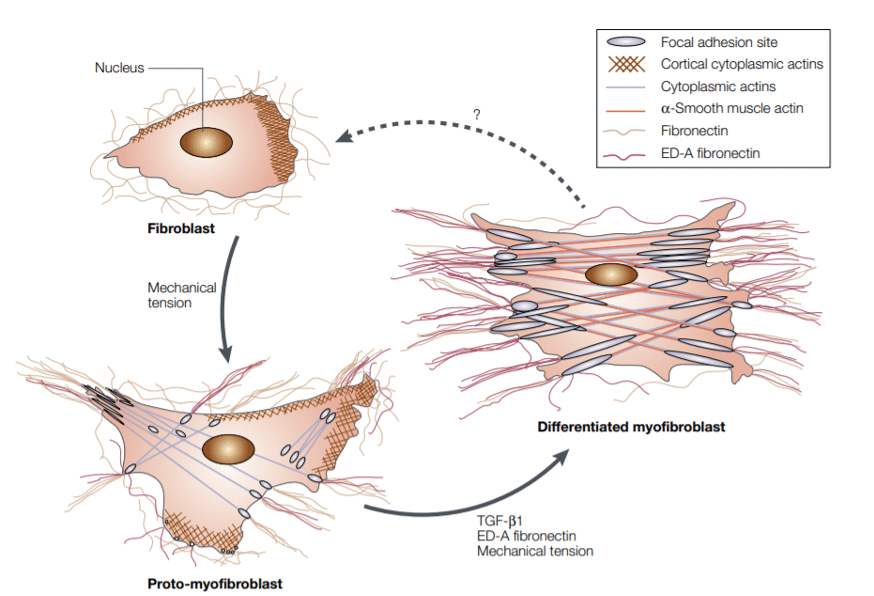
Figure 4. Model of fibroblast differentiation into myofibroblast
Tomasek et al. (2002)
Thus, contractility is an important feature of fibroblasts. On the one hand, it is an intrinsic ability, meaning that according to certain stimuli, they can contract and shape their surrounding environment. On the other hand, they are able to sense the forces applied to them and have specific molecular and cellular responses accordingly. There are therefore two important families of assays in the “contractility assays”: the study of the contractile forces generated by the cells, and the study of their response to a specific applied contraction.
> Signalling pathways
We have seen in the previous paragraph that the fibroblasts have the particular capacity to provide contractile forces. We will see in this part a more common feature, which is the ability to signal through the release of chemicals, and the ability to sense various kinds of signals and respond to them.
The cytokine transforming growth factor – β (TGF-β) is a polypeptide that is widely distributed in many tissues in the body and secreted by several types of cells, including macrophages, platelets, etc. (Raghow et al., 1987). Raghow et al. showed that TGF-β acts at a pre-translational level in human dermal fibroblasts. Indeed, the sensing of TGF-β resulted in the accumulation of mRNA for fibronectin and type I procollagen, and in an increasing amount of collagen production (Raghow et al., 1987). In their study, they used classical cell cultures for their experiments. They both quantified the amount of collagenous proteins using collagenase assays, and the transcription levels of collagen and fibronectin (levels of mRNA).
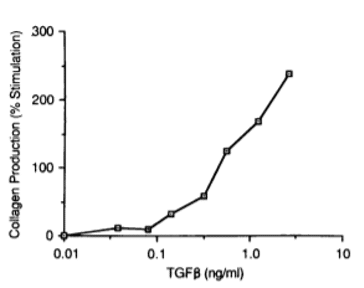
Figure 5. Collagen biosynthesis depending on the administration of TGF-β
Raghow et al. (1987)
In addition to triggering a change in transcription levels of some particular genes, TGF-β also acts as a chemoattractant for the fibroblasts (Postlethwaite et al., 1987). It was shown using blind-well modified Boyden chemotaxis chambers equipped with porous filters.
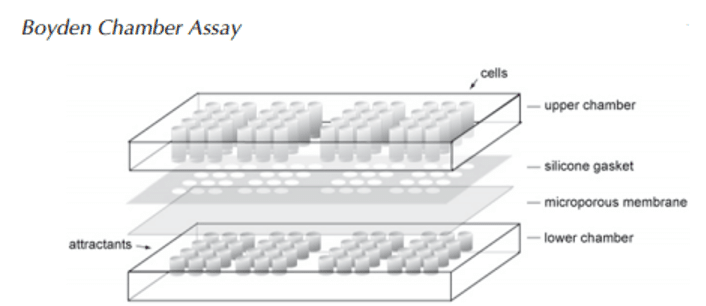
Figure 6. Boyden chemotaxis chamber
Guan (2004)
More generally, fibroblasts are able to sense a variety of cytokines, such as PDGF and IL-1, for example (Kohase et al., 1987). They are also able to produce a range of cytokines, like interferons (Kohase et al., 1987), IL-6 (Elias & Lentz, 1990), etc. In other words, fibroblast regulation comprises a complex cytokine network (which we will not develop exhaustively in this review, as it is not the main subject), allowing them to communicate with surrounding cells. The cytokine signals are present between different types of cells (paracrine manner), but also in an autocrine manner, meaning that the fibroblasts have the receptors for the cytokines they produce (Paulsson et al., 1987).
We will see later that these cytokine networks are of great importance for the fibroblasts, especially in the process of inflammation.
Before closing this section, we may have a word about the FGF (Fibroblast Growth Factor) and its role, as it carries the name “fibroblast”. Historically, it has been called FGF because it was discovered first as being a mitogen for fibroblasts (Gospodarowicz, 1974). Since then, researchers have determined that there are at least 22 members in the FGF family and that they act from embryonic development to adulthood in a variety of mechanisms, and on a wide variety of cells (Eswarakumar et al., 2005).
To sum up, in this section, we saw that the fibroblasts are regulated by a complex cytokine network and that they, in turn, produce various molecules which will regulate other types of cells. We will come back to these interactions in the second part of the review, which focuses on the role of fibroblasts in the body. Regarding the assays, in most of the papers cited above, the ways to measure the impact of the cytokines is to measure the mRNA levels or the protein levels of the molecule of interest on treated fibroblasts. We also reviewed the chemotaxis assay, which has been increasingly used, and it is becoming more sophisticated in order to mimic in the best way possible the real environment of the cells in the body. The chemotaxis assays are usually done in macroscopic chambers, with a membrane between the cell and the chemoattractant. Nevertheless, there are more sophisticated types of chambers. Such is the case of 4Dcell’s microfluidic chips to study chemotaxis. These devices are composed of two adjacent microfluidic chambers, connected by microchannels. The cells and chemoattractant are inserted in each one of the chambers, and a stable gradient is formed between the two of them. This gradient will cause the cells to migrate accordingly, enabling for example the study of their motility. It is a more sophisticated strategy to go from bulk to single cell analysis, in a 3D environment.
> Electrical function
It has been shown early on that certain cell lines of fibroblasts (L cells) are able to transmit a hyperpolarization signal of the membrane to neighbouring cells. The ranges given by Nelson and Peacock in 1973 are the following: a 20-30 mV increase in the membrane potential, which can be transmitted until a distance of approximately 0.5 mm (Nelson & Peacock, 1973). What they explained in their paper is that it is unlikely to be due to direct electrical conduction, or to ion exchange between neighbouring cells. They rather hypothesized that it is due to contact, by the mean of contraction and transmission of mechanical load.
The ability to transmit electrical signals may have various effects in the body, and we will discuss it especially when talking about the role of the fibroblasts in the heart.
> Synthesis of collagen and other ECM components
Collagen is the main protein in the human body, accounting for 25% to 35% of the whole-body content. It is the main protein in the ECM of the connective tissues, and the fibroblasts are the most common type of cells which are known to participate extensively in the production of this protein. They also produce other ECM proteins, such as glycoproteins and fibronectin (Baum et al., 1977).
Figure 7. Collagen triple helix
Shoulders and Raines (2009)
It has been shown in 1986 by Varga and Jimenez that TGF-β causes a significant increase in the production of collagen by human dermal fibroblasts (Varga & Jimenez, 1986), and by Usui et al. (1998) that TGF-β regulation of ECM production in human corneal fibroblasts is mediated especially by the Smad family of proteins (Usui et al., 1998). In this latest paper, researchers investigated their hypothesis using assays such as Western Blot, to detect the presence of proteins.
Fibroblasts not only have the ability to produce ECM components, but they also sense these proteins, and this interaction, in turn, influences their behaviour. Indeed, it has been shown that fibroblasts cultured in a 3D collagen gel structure produced more fibronectin as compared to the fibroblasts in monolayer cultures (Adachi et al., 1998).
These results have been obtained in part using 2D cell cultures, where chemicals are tested, and the level of a gene of interest is measured. The fact that the fibroblasts are able to sense the components of the ECM suggests that to get closer to the real physiological environment of the cells, we could use 3D collagen gels for the assays. 4Dcell developed a method to microstructure polyacrylamide based gels, being able to tune their different stiffness and functionalization according to the needs of the researchers.
> PROCESSES IN WHICH FIBROBLASTS ARE IMPLIED IN THE BODY
In the previous section of the review, we described some of the intrinsic properties of the fibroblasts. In this section, we are going to see how fibroblasts use these features to interact with other cells or tissues, and what functions they fulfil in the body.
> Tissue construction and modelling
One of fibroblasts’ critical roles is to contribute to tissue construction and modelling. We already saw that fibroblasts are responsible for the majority of collagen and other ECM components production. It gives structure and support to the intercellular space. It also constitutes the connective tissue in the organs. But the role of fibroblasts is not restricted to the production of ECM components, as they also actively support the formation and shaping of the tissues.
Using co-culture assays, it has been shown that fibroblasts release proliferation-stimulating soluble factors, which contribute to the good development of enough functional layers in the epidermis (El-Ghalbzouri et al., 2002). Indeed, keratinocytes seeded on collagen matrices with embedded fibroblasts showed good proliferation, differentiation, and epidermis morphogenesis, whereas keratinocytes seeded on collagen matrices without fibroblasts resulted on a stratified epidermis with only a few viable layers. The results are similar for the culture of keratinocytes with or without medium from fibroblast culture. These results support the fact that fibroblasts release some soluble factors which stimulate the proliferation of keratinocytes and promote the development of a functional epidermis.
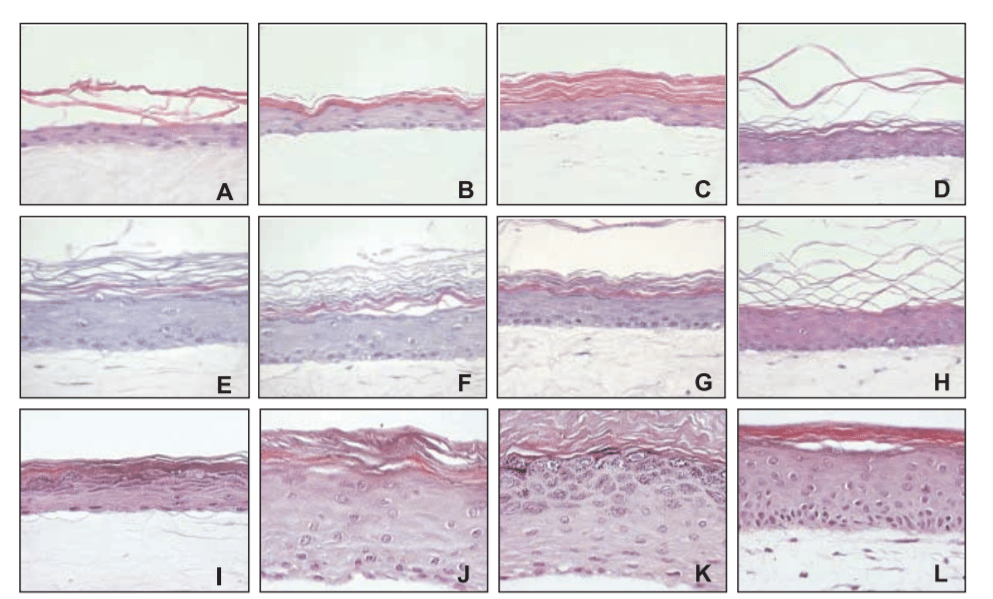
Figure 8. Reconstructed epidermis established on collagen matrices with increasing amounts of fibroblasts (A-H) or with medium derived from keratinocyte monocultures (I), dermal equivalent (J), keratinocyte-fibroblast co-culture (K-L)
El-Ghalbzouri et al. (2002)
In addition to promoting the good development of skin layers alongside other cells, fibroblasts also contribute directly to tissue modelling. We already saw that they are one of the major producers of collagen and other ECM components, but they are also responsible for the good alignment of these fibres. Back in 1982, Stopak et al. showed that the traction force they exert is sufficient to show patterns of contraction, distortion and alignment of collagen gels (Stopak & Harris, 1982). They proposed that this mechanism accounts for the good alignment and construction of tendons, ligaments and muscles during development.
Importantly, there is a diversity in fibroblast lineage, which reflects different functions in skin development and maintenance. Driskell et al. showed, for example, using lineage tracing experiments, that the fibroblasts from the upper and lower dermis come from different precursors, and that they fulfil different functions (Driskell et al., 2013). The upper dermis fibroblasts regulate hair growth, whereas the lower dermis fibroblasts comprise the reticular fibroblasts, which are major providers of fibrillar and bulk ECM components.
Thus, fibroblasts constitute a diverse group of cells which are responsible for the construction and modelling of the tissues. We saw examples in the skin, for which researchers used assays such as lineage tracing, reporter gene tracing (to assess the activity or proliferation state of keratinocytes), or co-culture assays on collagen matrices. This last type of assay is currently more and more developed, coherently with the wish to model as good as possible the physiological environment of cells. It is the spirit of 4Dcell to promote these kinds of technologies, and the 3D gels developed by the company would be suitable for the conduction of these types of experiments, providing different stiffness and composition according to the needs.
> Inflammation and immune response
We saw that among the features of fibroblasts, there is the complex cytokine network used to communicate with other fibroblasts, as well as with other types of cells. This is particularly relevant for their role in inflammation.
Smith et al. (1997) suggested in a commentary in the American Journal of Pathology that fibroblasts should be considered as sentinel cells (Smith et al., 1997). The first fact which supports their idea is the ability of the fibroblasts to be activated by inflammatory cytokines such as IL-1 and TNF-α, produced by macrophages upon sensing of lipopolysaccharides (LPS), which are components of the bacteria wall which are recognized by the macrophages upon bacterial infection (Rolfe et al., 1992). This interaction is crucial because after this activation, fibroblasts, as opposed to macrophages, are able to secrete monocyte chemoattractant protein-1 (MCP-1), a chemokine which initiates the recruitment of monocytes, and other hematopoietic cells. In other words, in this particular example, fibroblasts are the intermediate cells between different types of hematopoietic cells. They are also important in the adaptive response displayed by lymphocytes. Indeed, it has been shown that fibroblasts, previously activated by interferon-gamma (IFN-γ, another family of cytokines), interact with infiltrating T lymphocytes (via CD40 expression) to promote inflammation and fibrogenesis. The interaction seems to go both ways, as circulating CD40 activates further the fibroblasts (Sempowski et al., 1997).
Moreover, as players of the immune response (innate and adaptive), fibroblasts also have the ability to moderate the inflammatory response, thus avoiding an overreaction, which is detrimental to the organism. In fact, in fibroblasts, RelB (a transcription factor) helps to shut down the expression of cytokines and chemokines (Xia et al., 1997). For relatively short-lived cells such as macrophages, these regulatory mechanisms are of less importance because the cytokine production is arrested by cell death. For fibroblasts, which are long-lived cells, these mechanisms are absolutely required, as shown in vitro and in vivo (Xia et al., 1997).
Therefore, fibroblasts are important cells for the immune response, as they are among the first activated cells, and contribute to the recruitment of other hematopoietic cells. The papers cited above used in vitro and in vivo techniques such as knockdown assays and stimulation assays. Given the complexity of the interactions between all the cells in play, it would be interesting to reach a novel step, and study them together, in a 3D environment, rather than separated. 4Dcell products such as microfluidic devices can be used to study chemotaxis. On the other hand, the micropatterned based substrates can be used as co-culture assays.
> Wound healing
In wound healing, there are three critical phases, these being inflammation, proliferation, and remodelling. We already saw the implication of fibroblasts during inflammation, and the role they play in the production of ECM and its modelling.
Another important feature during wound healing is the contraction of the wound, which ultimately leads to its closure. The activation of the fibroblasts and differentiation into myofibroblasts, which express α-SMA, is thought to trigger wound contraction and closure (Tomasek et al., 2002). These features are the ones we described in the first part, in the subsection about contractility.
Mechanical tension itself seems to be important for the differentiation of fibroblasts into myofibroblasts (Meyer-ter-Vehn et al., 2006). Once the fibroblasts are stimulated with TGF-β, the cellular tension increases, ultimately leading to the formation of α-SMA networks and differentiation into myofibroblasts. When they blocked the cellular tensions (using a small molecule inhibitor: Y-27632), researchers observed poor differentiation rate and poor scarring. They suggested targeting this mechanism as a therapeutic route in the modulation of detrimental scarring (glaucoma surgery in their case).
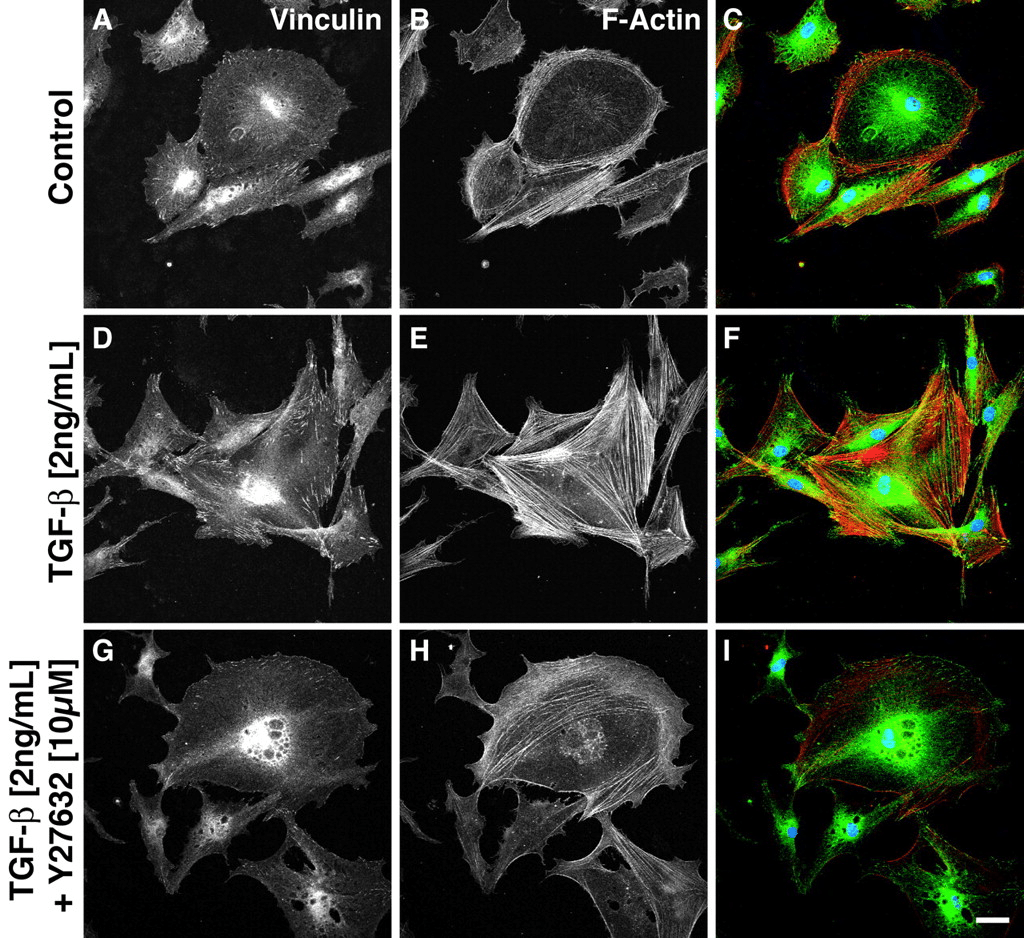
Figure 9. Fibroblasts were stimulated with vehicle (A-C), TGF-β alone (D-F), or TGF-β with Y-27632 (blockage of cellular tension), and stained for vinculin (A, D, G) and actin (B, E, H). Composite images (C, F, I) show vinculin in green, actin in red, and the nucleus in blue (DAPI staining)
Meyer-ter-Vehn et al. (2006)
Myofibroblasts are of great importance for wound contraction and closure. They are present in the granulation tissue, which is a transitory tissue, particularly rich in myofibroblasts and vascular cells. During the transition between granulation tissue and scar, there is a loss in the number of myofibroblasts and vascular cells. Desmoulière et al. (1995) suggested, given the results they obtained in their study, that these cells are cleared by apoptosis (Desmoulière et al., 1995).
An assay that, although still under development, is increasingly being utilised is the “scratch assay (Liang et al., 2007). The principle is that, in a monolayer cell culture, we make a scratch, and acquire images before and periodically after the scratch. It is extensively used to study, for example, cell migration, but it can also be seen as a tool to obtain images from myofibroblasts during this artificial wound closure. 4Dcell dynamic micropatterns allow the same kind of experiments, without the interference of the researcher making the scratch. Indeed, the micropatterns are a collection of adhesive and antiadhesive spaces, and the cells are seeded on top of the adhesive ones. At a certain time (the time at which we would do the scratch ourselves in the scratch assay), the anti-adhesive space can be rendered adhesive, and it will allow the researchers to see the myofibroblasts repairing this artificial wound.
> Fibrosis and pathological wound healing
We have seen in the previous paragraph, the process of wound healing and how fibroblasts are involved in this mechanism. After the wound is healed, there is a scar, which is the normal ending of the process. If the healing goes awry and doesn’t stop, there is emergence of a fibrotic scarring, which comprises considerable tissue remodelling, and formation of a permanent scar tissue (Wynn, 2004). Even if the fibrotic diseases are very diverse, they have in common a persistent inflammatory state, rich in lymphocytes and monocytes which interact together, and sustain the production of growth factors and fibrogenic chemicals. Together, they promote the production of exacerbated connective tissue matrix, which takes precedence over the normal tissue, resulting ultimately in organ failure and death.
Fibrotic diseases are observed in many organs, especially in the lungs (idiopathic pulmonary fibrosis), heart (cardiac fibrosis), kidney (chronic kidney disease), and liver (cirrhosis).
In the case of patients with idiopathic pulmonary fibrosis (IPF), the conventional treatment has been corticosteroids for a while, because they have an anti-inflammatory effect (Scotton & Chambers, 2007). But this treatment still leaves the patient with poor prognosis, and the survival rate after five years doesn’t exceed 20%. Another therapeutic axis is the administration of anti-fibrotic agents, such as pirfenidone (blocks collagen synthesis, extracellular matrix secretion and fibroblast proliferation) (Gross & Hunninghake, 2001). The clinical studies showed a slight improvement for the patients upon treatment with pirfenidone (King et al., 2014). In the process of fibrosis, the major players are the myofibroblasts, which are also the effector cells during wound healing (they can generate contractile forces and are major producers of ECM components). As control of the inflammation isn’t a therapeutic success with the patients progressively losing the lung function, researchers tried to find a way to target the myofibroblasts directly (Scotton & Chambers, 2007). The study of myofibroblasts in IPF is thus particularly relevant.
In the heart, the functional cells are the cardiomyocytes, embedded in a collagen-rich matrix, with a precise tensile force, and the stability is ensured in part by the fibroblasts (Weber, 1989). We will go deeper into the functional fibroblast-cardiomyocyte interaction in the heart in the next paragraph. If the cardiomyocytes are lost because of any kind of heart injury, the fibroblasts get activated, differentiate into myofibroblasts, and mount a healing response. The incorporation and modelling of type I collagen ensure the structural integrity of the heart (Weber et al., 2013). When the myofibroblasts don’t respond to apoptotic signals at the end of the healing process, they perpetually produce connective tissue matrix, resulting in the development of fibrosis. At this point, the architecture of the heart gets disrupted.
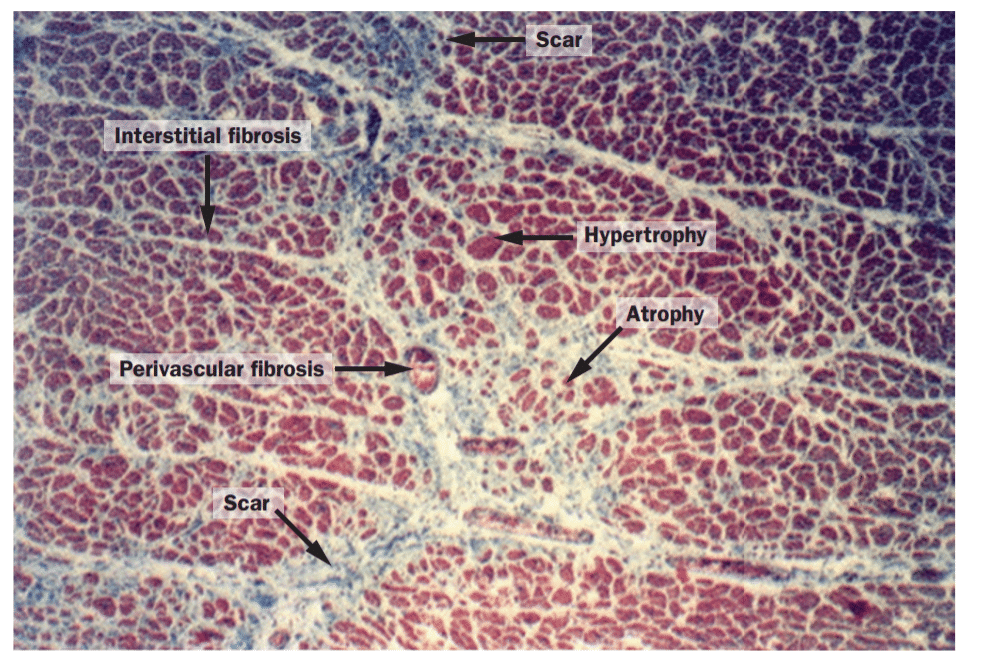
Figure 10. Myocardium (post mortem tissue) in hypersensitive heart disease
Weber et al. (2013)
In this part, we showed an important role for fibroblasts, and more precisely myofibroblasts, in the onset of fibrotic diseases. In the paper quoted above, the researchers mainly used 2D cell cultures (especially for the testing of molecules for therapeutics), and animal models, to be able to study the histology of the tissues under various conditions. 3Dgel technology provided by 4Dcell are at the interface between classical 2D cell culture and in vivo studies, as it reproduces more accurately the physiological environment, leading ultimately to the reduction of the number of animals used. Indeed, the tuneable 3D environment provided by the gels would enable the researchers to narrow down the candidate molecules, before moving to studies with animal models.
> Fibroblasts and cardiomyocytes
In the previous subsection, we saw that in the heart, fibroblasts intervene when the cardiomyocytes are lost, so that the function of the heart is not compromised (Weber et al., 2013). But above the wound healing processes, fibroblasts are also important in the interaction with cardiomyocytes in normal and non-wounding periods.
If the fibroblasts have first been considered as unexcitable cells which mainly act as an insulator between different groups of cardiomyocytes in the heart, it seems that they can also influence the excitability of myocytes and, therefore, also influence impulse propagation. Indeed, Miragoli et al. (2007) used ventricular cardiomyocytes coated with a low number of (or without) myofibroblasts to show that the cardiomyocytes exhibit spontaneous activity in a density-dependent manner (myofibroblasts dependent), whereas the cardiomyocytes alone remained quiescent (Miragoli et al., 2007).
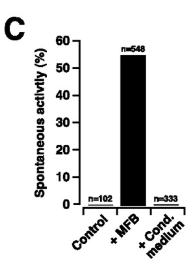
Figure 11. Spontaneous activity of the cardiomyocytes according to culture conditions
Miragoli et al. (2007)
The picture above, from Miragoli et al. (2007), shows the spontaneous activity of the cardiomyocytes alone (control), with myofibroblasts (+MFB) or with myofibroblasts conditioned medium (+cond medium).
These findings show the need of culture systems which are closer to the physiological environment of the cells, in terms of 3D shape, of matrix, but also of neighbouring cells. In this context, more and more assays using co-cultures are currently being developed, representing one of the current topics 4Dcell is working on.
> CONCLUSION
We saw that fibroblasts are a very particular type of cell. They are mesenchymal cells, meaning that as opposed to parenchymal cells, they are not the functional cells which ensure the functionalities of the organs. Nevertheless, they provide support and structure to the functional cells in most of the tissues of the human body. They are the major cells responsible for the production and modelling of ECM components, and are the cells in charge of repairing it in case there is an injury. Like other types of cells, fibroblasts need to be regulated, and an excess in fibroblast activation and action can lead to fibrosis and, ultimately, organ failure or death.
As they are present in most tissues, and lie in the connective tissue, they are among the first cells in line when there is an injury or an infection. As such, they also play a very important role in inflammation and immune response, through various cytokine networks.
Because of their broad implication in the body, and their various types of functions, they are extensively studied. Classical 2D assays in which we apply a stimulus and measure a response are of great interest to the basic comprehension of the mechanisms of the cells. We also describe that new forms of assays, in 3D and eventually in co-culture with other cell types, are being developed for the past years. As examples, we can quote the collagen matrices used for cell culture and the co-cultures with cardiomyocytes, which enabled the researchers to discover the stimulating potential of the fibroblasts. 4Dcell mission is to provide innovative devices which allow us to control the cell microenvironment, thus getting closer to the physiological environment of cells. These devices are also thought of as smart tools enabling the measurement of physical quantities, like cellular migration speed, as well as enabling researchers to acquire images and study the mechanisms at a single cell level (using 3D gels, microchannels or micropatterns).
> REFERENCES
Adachi, Y., Mio, T., Takigawa, K., Striz, I., Romberger, D. J., Spurzem, J. R., & Rennard, S. I. (1998). Fibronectin production by cultured human lung fibroblasts in three-dimensional collagen gel culture. In Vitro Cellular & Developmental Biology – Animal, 34(3), 203–210. https://doi.org/10.1007/s11626-998-0125-7
Balaban, N. Q., Schwarz, U. S., Riveline, D., Goichberg, P., Tzur, G., Sabanay, I., Mahalu, D., Safran, S., Bershadsky, A., Addadi, L., & Geiger, B. (2001). Force and focal adhesion assembly: a close relationship studied using elastic micropatterned substrates. Nature Cell Biology, 3(5), 466–472. https://doi.org/10.1038/35074532
Baum, B. J., McDonald, J. A., & Crystal, R. G. (1977). Metabolic fate of the major cell surface protein of normal human fibroblasts. Biochemical and Biophysical Research Communications, 79(1), 8–15. https://doi.org/10.1016/0006-291X(77)90053-5
Bereiter-Hahn, J. (2005). Mechanics of crawling cells. Medical Engineering & Physics, 27(9), 743–753. https://doi.org/10.1016/j.medengphy.2005.04.021
Desmoulière, A., Redard, M., Darby, I., & Gabbiani, G. (1995). Apoptosis mediates the decrease in cellularity during the transition between granulation tissue and scar. The American Journal of Pathology, 56–66.
Driskell, R. R., Lichtenberger, B. M., Hoste, E., Kretzschmar, K., Simons, B. D., Charalambous, M., Ferron, S. R., Herault, Y., Pavlovic, G., Ferguson-Smith, A. C., & Watt, F. M. (2013). Distinct fibroblast lineages determine dermal architecture in skin development and repair. Nature, 504(7479), 277–281. https://doi.org/10.1038/nature12783
El-Ghalbzouri, A., Gibbs, S., Lamme, E., Van Blitterswijk, C. A., & Ponec, M. (2002). Effect of fibroblasts on epidermal regeneration. British Journal of Dermatology, 147(2), 230–243. https://doi.org/10.1046/j.1365-2133.2002.04871.x
Elias, J. A., & Lentz, V. (1990). IL-1 and tumor necrosis factor synergistically stimulate fibroblast IL-6 production and stabilize IL-6 messenger RNA. The Journal of Immunology, 145, 161–166.
Eswarakumar, V. P., Lax, I., & Schlessinger, J. (2005). Cellular signaling by fibroblast growth factor receptors. Cytokine & Growth Factor Reviews, 16(2), 139–149. https://doi.org/10.1016/j.cytogfr.2005.01.001
Fibroblast. (n.d.). In Encyclopaedia britannica.
Gospodarowicz, D. (1974). Localisation of a fibroblast growth factor and its effect alone and with hydrocortisone on 3T3 cell growth. Nature, 249(5453), 123–127. https://doi.org/10.1038/249123a0
Grinnell, F., Ho, C.-H., Lin, Y.-C., & Skuta, G. (1999). Differences in the Regulation of Fibroblast Contraction of Floating Versus Stressed Collagen Matrices. Journal of Biological Chemistry, 274(2), 918–923. https://doi.org/10.1074/jbc.274.2.918
Gross, T. J., & Hunninghake, G. W. (2001). Idiopathic Pulmonary Fibrosis. New England Journal of Medicine, 345(7), 517–525. https://doi.org/10.1056/NEJMra003200
Guan, J.-L. (2004). Cell Migration (Vol. 294). Humana Press. https://doi.org/10.1385/1592598609
Harris, A., Wild, P., & Stopak, D. (1980). Silicone rubber substrata: a new wrinkle in the study of cell locomotion. Science, 208(4440), 177–179. https://doi.org/10.1126/science.6987736
Hinz, B., Celetta, G., Tomasek, J. J., Gabbiani, G., & Chaponnier, C. (2001). Alpha-Smooth Muscle Actin Expression Upregulates Fibroblast Contractile Activity. Molecular Biology of the Cell, 12(9), 2730–2741. https://doi.org/10.1091/mbc.12.9.2730
Hinz, B., Mastrangelo, D., Iselin, C. E., Chaponnier, C., & Gabbiani, G. (2001). Mechanical Tension Controls Granulation Tissue Contractile Activity and Myofibroblast Differentiation. The American Journal of Pathology, 159(3), 1009–1020. https://doi.org/10.1016/S0002-9440(10)61776-2
Kalluri, R., & Zeisberg, M. (2006). Fibroblasts in cancer. Nature Reviews Cancer, 6(5), 392–401. https://doi.org/10.1038/nrc1877
King, T. E., Bradford, W. Z., Castro-Bernardini, S., Fagan, E. A., Glaspole, I., Glassberg, M. K., Gorina, E., Hopkins, P. M., Kardatzke, D., Lancaster, L., Lederer, D. J., Nathan, S. D., Pereira, C. A., Sahn, S. A., Sussman, R., Swigris, J. J., & Noble, P. W. (2014). A Phase 3 Trial of Pirfenidone in Patients with Idiopathic Pulmonary Fibrosis. New England Journal of Medicine, 370(22), 2083–2092. https://doi.org/10.1056/NEJMoa1402582
Kohase, M., May, L. T., Tamm, I., Vilcek, J., & Sehgal, P. B. (1987). A cytokine network in human diploid fibroblasts: interactions of beta-interferons, tumor necrosis factor, platelet-derived growth factor, and interleukin-1. Molecular and Cellular Biology, 7(1), 273–280. https://doi.org/10.1128/MCB.7.1.273
Kolodney, M., & Wysolmerski, R. (1992). Isometric contraction by fibroblasts and endothelial cells in tissue culture: a quantitative study. The Journal of Cell Biology, 117(1), 73–82. https://doi.org/10.1083/jcb.117.1.73
Li, B., & Wang, J. H.-C. (2011). Fibroblasts and myofibroblasts in wound healing: Force generation and measurement. Journal of Tissue Viability, 20(4), 108–120. https://doi.org/10.1016/j.jtv.2009.11.004
Liang, C.-C., Park, A. Y., & Guan, J.-L. (2007). In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nature Protocols, 2(2), 329–333. https://doi.org/10.1038/nprot.2007.30
Meyer-ter-Vehn, T., Sieprath, S., Katzenberger, B., Gebhardt, S., Grehn, F., & Schlunck, G. (2006). Contractility as a Prerequisite for TGF-β–Induced Myofibroblast Transdifferentiation in Human Tenon Fibroblasts. Investigative Opthalmology & Visual Science, 47(11), 4895. https://doi.org/10.1167/iovs.06-0118
Miragoli, M., Salvarani, N., & Rohr, S. (2007). Myofibroblasts Induce Ectopic Activity in Cardiac Tissue. Circulation Research, 101(8), 755–758. https://doi.org/10.1161/CIRCRESAHA.107.160549
Nelson, P. G., & Peacock, J. H. (1973). Transmission on an Active Electrical Response between Fibroblasts (L Cells) in Cell Culture. The Journal of General Physiology, 62, 25–36.
Paulsson, Y., Hammacher, A., Heldin, C.-H., & Westermark, B. (1987). Possible positive autocrine feedback in the prereplicative phase of human fibroblasts. Nature, 328(6132), 715–717. https://doi.org/10.1038/328715a0
Postlethwaite, A. E., Keski-Oja, J., Moses, H. L., & Kang, A. H. (1987). Stimulation of the chemotactic migration of human fibroblasts by transforming growth factor beta. The Journal of Experimental Medicine, 165(1), 251–256. https://doi.org/10.1084/jem.165.1.251
Raghow, R., Postlethwaite, A. E., Keski-Oja, J., Moses, H. L., & Kang, A. H. (1987). Transforming growth factor-beta increases steady state levels of type I procollagen and fibronectin messenger RNAs posttranscriptionally in cultured human dermal fibroblasts. Journal of Clinical Investigation, 79(4), 1285–1288. https://doi.org/10.1172/JCI112950
Rolfe, M. W., Kunkel, S. L., Standiford, T. J., Orringer, M. B., Phan, S. H., Evanoff, H. L., Burdick, M. D., & Strieter, R. M. (1992). Expression and regulation of human pulmonary fibroblast-derived monocyte chemotactic peptide-1. American Journal of Physiology-Lung Cellular and Molecular Physiology, 263(5), L536–L545. https://doi.org/10.1152/ajplung.1992.263.5.L536
Scotton, C. J., & Chambers, R. C. (2007). Molecular Targets in Pulmonary Fibrosis. Chest, 132(4), 1311–1321. https://doi.org/10.1378/chest.06-2568
Sempowski, G. D., Chess, P. R., & P, P. R. (1997). CD40 is a functional activation antigen and B7-independent T cell costimulatory molecule on normal human lung fibroblasts. The Journal of Immunology, 4670–4677.
Shoulders, M. D., & Raines, R. T. (2009). Collagen Structure and Stability. Annual Review of Biochemistry, 78(1), 929–958. https://doi.org/10.1146/annurev.biochem.77.032207.120833
Smith, R. ., Smith, T. ., Blieden, T. ., & Phipps, R. . (1997). Fibroblasts as sentinel cells. Synthesis of chemokines and regulation of inflammation. The American Journal of Pathology, 151, 317–322.
Stopak, D., & Harris, A. K. (1982). Connective tissue morphogenesis by fibroblast traction. Developmental Biology, 90(2), 383–398. https://doi.org/10.1016/0012-1606(82)90388-8
Tomasek, J. J., Gabbiani, G., Hinz, B., Chaponnier, C., & Brown, R. A. (2002). Myofibroblasts and mechano-regulation of connective tissue remodelling. Nature Reviews Molecular Cell Biology, 3(5), 349–363. https://doi.org/10.1038/nrm809
Usui, T., Takase, M., Kaji, Y., Ishida, K., Tsuru, T., Miyata, K., Kawabata, M., & Yamashita, H. (1998). Extracellular matrix production regulation by TGF-beta in corneal endothelial cells. Investigative Ophtalmology & Visual Science, 39(11), 1981–1989.
Varga, J., & Jimenez, S. A. (1986). Stimulation of normal human fibroblast collagen production and processing by transforming growth factor-β. Biochemical and Biophysical Research Communications, 138(2), 974–980. https://doi.org/10.1016/S0006-291X(86)80591-5
Weber, K. T. (1989). Cardiac interstitium in health and disease: The fibrillar collagen network. Journal of the American College of Cardiology, 13(7), 1637–1652. https://doi.org/10.1016/0735-1097(89)90360-4
Weber, K. T., Sun, Y., Bhattacharya, S. K., Ahokas, R. A., & Gerling, I. C. (2013). Myofibroblast-mediated mechanisms of pathological remodelling of the heart. Nature Reviews Cardiology, 10(1), 15–26. https://doi.org/10.1038/nrcardio.2012.158
Wynn, T. A. (2004). Fibrotic disease and the TH1/TH2 paradigm. Nature Reviews Immunology, 4(8), 583–594. https://doi.org/10.1038/nri1412
Xia, Y., Pauza, M. E., Feng, L., & Lo, D. (1997). RelB regulation of chemokine expression modulates local inflammation. The American Journal of Pathology, 375–387.