> MAKE YOUR NEURONS HAPPIER!
Control, guide and evaluate outgrowth of neuronal culture
> MAKE YOUR NEURONS HAPPIER!
Control, guide and evaluate outgrowth of neuronal culture
Neurons are often cultured in vitro on a flat, open, and rigid substrate, a platform that does not reflect well the native microenvironment of the brain. The cell’s microenvironment is defined by chemical and mechanical parameters. In conventional cell cultures, these environmental parameters could be controlled for a population of cells, but cannot be addressed locally to individual cells. One of the major interests of microfluidics is environmental control at the scale of the cell (Velve-Casquillas et al., 2010)
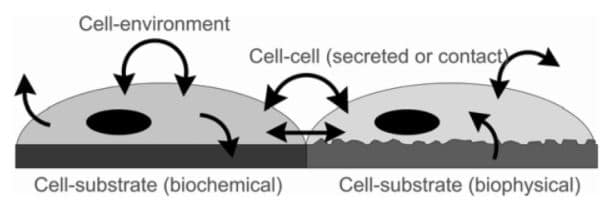
Local signals regulate cell behavior. Cellular processes such as adhesion, migration, growth, secretion, and gene expression are triggered, controlled, or influenced by local biophysical and biochemical signals (Li et al., 2003).
> GEOMETRICAL CONTROL OF CELL MICROENVIRONNEMENT: SIZE & DIRECTION OF NEURONAL CULTURE
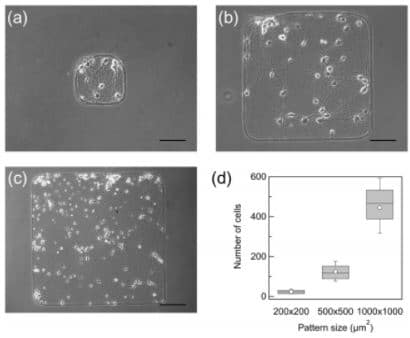
Synchronization of primary rat cortical neurons depends on the network size on micropatterns. Micropatterns used to restrict the area of cultured cortical network were (a) 200 × 200 μm2, (b) 500 × 500 μm2, and (c) 1000 × 1000 μm2 (Yamamoto et al., 2016).
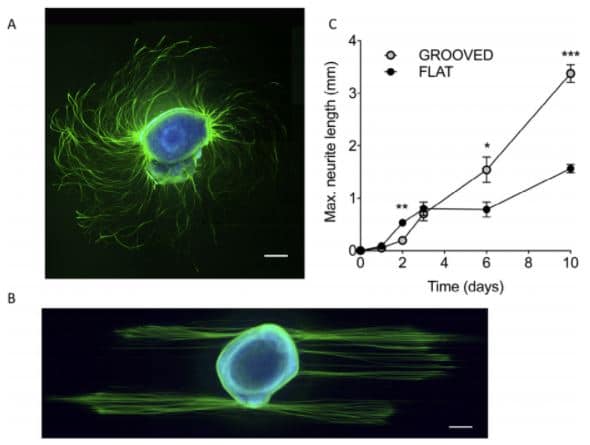
Neurite guidance: cues microfabricated surfaces. The dorsal root ganglion explant was cultured for 72h on flat (A) or patterned (B) PLL coated PDMS substrates. Labelling: ß3-tubulin (green) and DNA (blue). A) outgrowth on a flat substrate (turning spiral arrangement of axons); B) The topographical cues direct neurite outgrowth. Scale bars 200 µm. C) Plot of maximum neurite length versus time (Thomson et al., 2017).
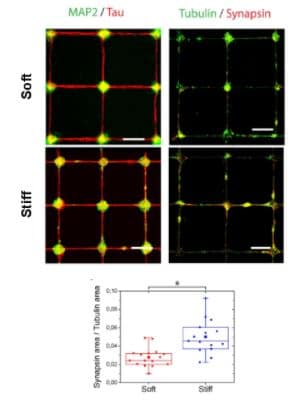
Modulation of the assembly dynamics and the functional activity of cortical neurons by tuning matrix stiffness and surface patterning.
Epifluorescent images of cortical neuronal networks grown on soft and stiff surfaces and immunostained for MAP2/Tau and βIII-tubulin/synapsin. The ratio of synapsin/tubulin areas was quantified in NIS Elements Advanced Research (v4.5, Nikon, Japan)(Lantoine et al., 2016).
> CELL-CELL INTERACTION, SYNAPSE FORMATION, AND COLLISION ASSAY
In a neuronal network, signal transmission between neurons occurs at specialized contact sites : the synapses. Primary cultures of dissociated neurons in vitro are capable of forming functional synapses (Fletcher et al. 1991, Fletcher et al. 1994). However, experiments with complex neural circuits do not allow for control over a single synaptic contact.
Engineered surfaces have shown promise as a powerful tool to investigate cell-cell communication at a synaptic level. With the use of micropatterns, neuronal networks investigation has expanded its knowledge in the field of neurosciences.
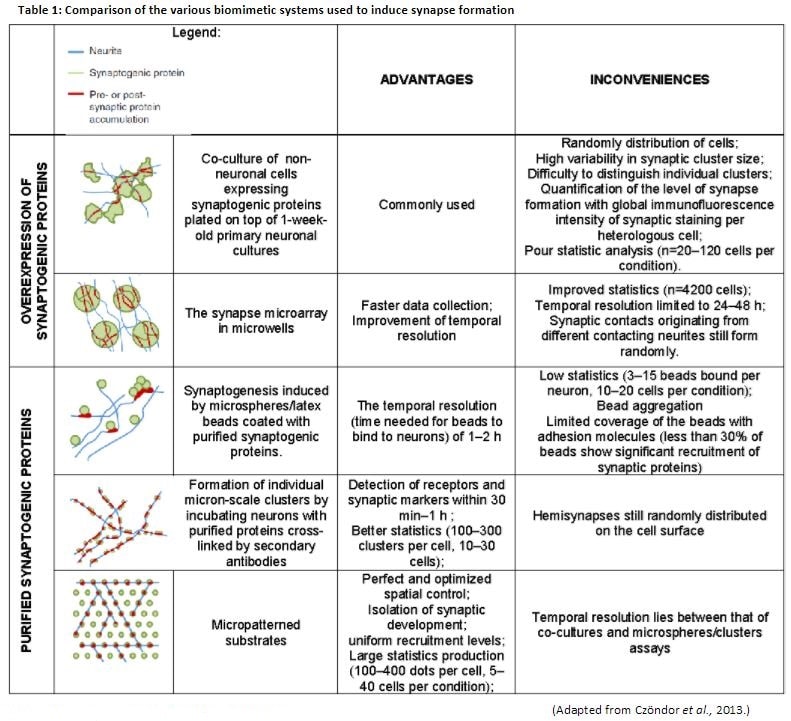
Substrate adhesion, a key parameter that determines the growth and survival of neurons in vitro, is governed by a number of factors. Several experimental designs were elaborated to induce and to study synapse formation. However, the strong advantage of micropatterned substrates over such experiments is that one precisely controls the position where to expect synapse formation (Czöndor et al., 2013) (Table 1).
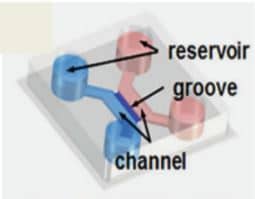
> CONTROL MICROENVIRONMENT IN 3D CONTEXT WITH MICROCHANNELS
Chips include inlets, as microwells, for cell body confinement, and microchannels capable of guiding neuritis for network topology specification.
In this context, the dimensions of the microchannels seemed to play a critical role. In fact, experiments with different channel dimensions showed that smaller channels of width and height of 5-10 µm yield better guidance than larger ones of 30 µm width and height (Krumpholz et al., 2015).
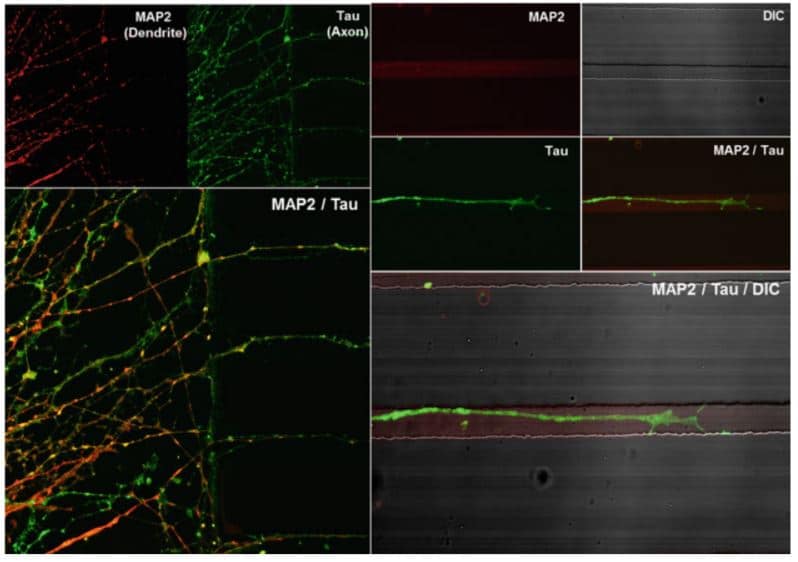
Monitor the differentiation and migration of neural cells derived from human embryonic stems cells
Single cells are seeded in reservoirs. Neurospheres will form in the channel and differenciate in the grooves (Bae et al., 2016). Dendrites and axons are visualized by immunostaining against MAP2 (red) and Tau (green). Only Tau positive axons are detected in the microchannels.
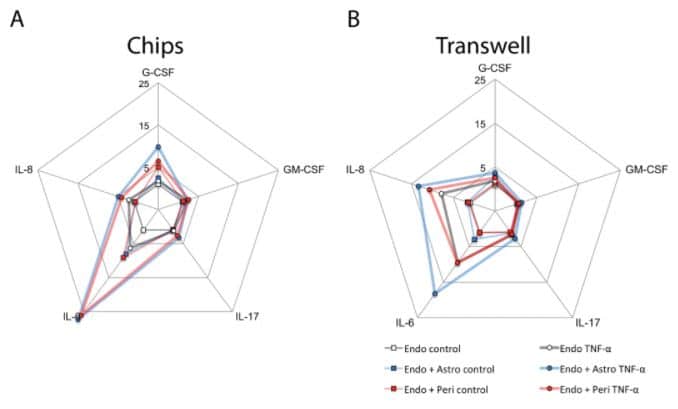
Organ-in-a-chip : 3D blood-brain barrier model
Using 3D human BBB-on-a-chip to study the neuroinflammatory response in vitro could be more relevant than classical culture system as mono/co-cultures in Transwell culture plates. Significant differences in cytokine and interleukine secretions by astrocytes and pericytes were observed (Herland et al., 2016).
Data were normalized to the levels of cytokines released by endothelial cells cultured alone; concentric scales indicate fold increase (Herland et al., 2016).
Keywords, Keypoints:
Neuronal circuit, neurons, neurite guidance, synapse formation, micropatterns, microfluidic confinment, microchannel, geometrical control, 2D/3D cell culture.