> CARDIAC ORGANOID – CARDIOTOXICITY
New technologies of engineered heart tissues for the prediction of cardiototoxicity during preclinical studies
New technologies of engineered heart tissues for the prediction of cardiototoxicity during preclinical studies
> Introduction
Drug safety is among the main concerns of pharmaceuticals who fight to improve it. In fact, both the long-term and the high costs of drug development lead to an urgent improvement in productivity. Many studies have shown that cardiotoxicity is one of the main causes leading to drug withdrawal (Stevens et al., 2009). By predicting the safety of the molecules tested on relevant models, risk of drug withdrawal will decrease.
In this sense, 4Dcell develops new technologies where the 3Rs strategy (Replacement, Reduction, Refinement) is promoted decreasing the amount of animal studies and consequently the ethical issues due to the use of animal models (Langley et al., 2015).
This review describes the state of the art of cardiotoxicity in vitro models and related technologies.
> Cell models
> Primary cells
Considered as a good model for cardiac research, rodent cardiomyocytes are often used for drug development (Li et al., 2014). Cell cultures allow to quantify and to identify cardiomyocytes functions, such as, electrophysiology, contractile mechanics or protein expression (Louch et al., 2011). However, they differ a lot from human cardiomyocytes and they are not able to be used indefinitely (Novakovic et al., 2014) leading to the development of new cell models for cardiac research.
Research using human pluripotent stem cells derived cardiomyocytes (hPS-CM) has been developed due to their similarities to human fetal cardiomyocytes regarding their contractility properties or maturation process (Snir et al., 2003).
Considered as the first pluripotent stem cell source, embryonic stem cells isolated from blastocytes are used in different research fields including drug development (Thomson et al., 1998) because they can form all somatic tissues. Moreover, in contrast to primary cells, they have an unlimited lifespan allowing long term studies. However, for ethical reasons, the use of ESC can be very contested. In fact, although those cells are greatly beneficial for clinical research, the use is discussed because they are derived from pre-implanted human embryos (Wert et al., 2003).
Nowadays, induced pluripotent stem cells (iPSC), obtained applying pluripotency factors on somatic cells, are used to counteract the negative effects of ESC and to obtain a better in vitro cell model for research (Takahashi et al., 2006). Comparing iPSC-CMs to cardiomyocytes from rats, it has been shown that the main cardiac proteins were detected in iPSC-CMs. Using those cells, adult cardiac cells with disorders can be obtained allowing the study of cellular phenotype and may undergo genetic modifications (Zuppinger et al., 2017).
> Cell culture systems
> Microstructured matrices
Micropatterned cell culture substrates are composed by adherent (fibronectin, collagen, etc.) and non-adherent areas, where the shape can vary from lines, rectangles, triangles, etc. When cultured in this type of devices, cells acquire the shape of adhesive areas.
Micropatterned matrices have been used to obtain an organized cell culture of cardiomyocytes, allowing the cells to develop their biological functions, which mimics in vivo conditions (Carter, 1967). For example, when cardiomyocytes are cultured in line-shaped patterns (Dussaud et al., 2018) (Figure 3), they become elongated, which improves sarcomere formation (Salick et al., 2014) and contractility (Ribeiro et al., 2015).
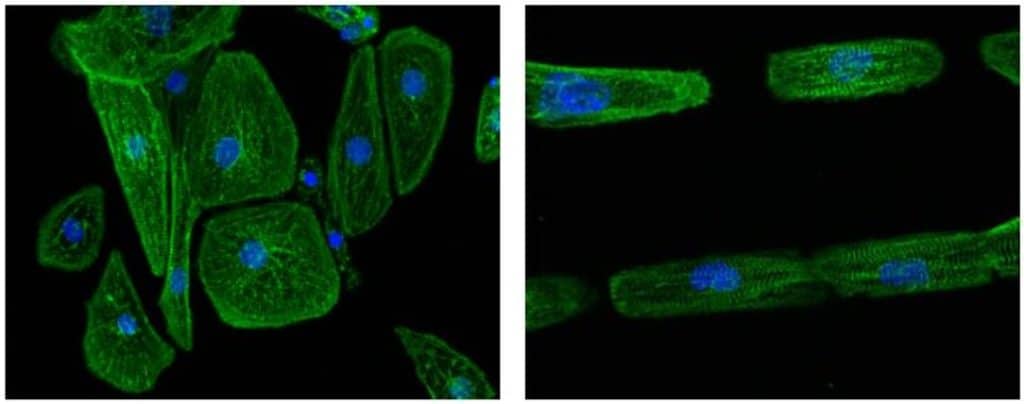
Figure 1. Images of unpatterned (left image) and line-micropatterned (right image) hPSC-CM
Dussaud S., Jouve C., Hulot J.S., 2018 (obtained using 4Dcell micropatterns)
In addition, gels can be used as a support for CMs culture, leading to a model that mimics native heart tissue (Tiburcy et al., 2014). It allows the cells to be assembled in a more organized fashion, thus improving maturation. For instance, alginate scaffolds have been made to enhance structural, functional and phenotypic properties allowing electrical communication between cells (Dvir et al., 2011).
> Hydrogels
One of the first idea of the engineered heart tissue (EHT) was to encapsulate cells into hydrogels in the 1980s using collagen gels (Weinberg et al., 1986). Then, diverse gel types have been developed such as Matrigel or fibrin. Made of three components (gel natural product, anchors and molds), it enables to the cell culture to grow in a 3D space and to be fixed. The main advantage of hydrogels is the ability of cells to spread and to interact between each other. The easy way to perform studies with this technology lead to the regular use of hydrogels especially for cardiac research (Hirt et al., 2014). Cardiac cells cultured with hydrogel are either on the top of it (Eschenhagen et al., 2017) or in microstructured shapes (Zimmerman et al., 2002). So, this technique is still improved in combination to other technologies to obtain the closest representation of in vivo conditions.
> Cellular self-organization: cell sheets and spheroid
Cell sheets
Usually, when cultured on plastic dishes, monolayers of cardiomyocytes are observed after some time culture. To keep the monolayers intact, cell sheets technique has been developed with a temperature-sensitive method to enable 3D tissue formation (Figure 2) at room temperature allowing beating of cells or electrical communication (Shimizu et al., 2002).
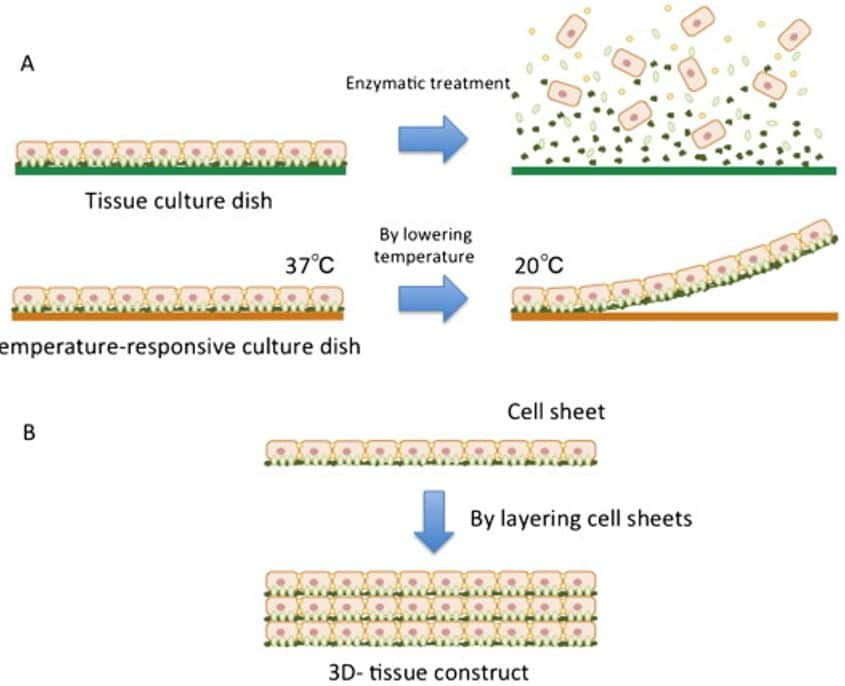
Figure 2. Principle of cell sheet technology.
A. using a temperature sensitive method, a cell sheet is formed. B. Layers of cell sheets enables a 3D-tissue formation (Masuda; Shimizu, 2016)
Spheroid
Spheroids are an alternative to 2D cell cultures, where cells are organized in 3D, enabling them to interact with surrounding cells and ECM, better mimicking in vivo conditions (Yan et al., 2018).
Spheroid can be formed from primary or iPSC-derived cardiac cells using, for example, the hanging drop method. This can be achieved by the inversion of the plates where the cells are suspended enabling the formation of drops. Thus leading to the formation of cell aggregates (Polonchuk et al., 2017; Kelm et al., 2003) (Figure 3).
In this conformation, cellular and molecular composition are reproduced like in cardiac tissue with a functional vascular network (Polonchuk et al., 2017).
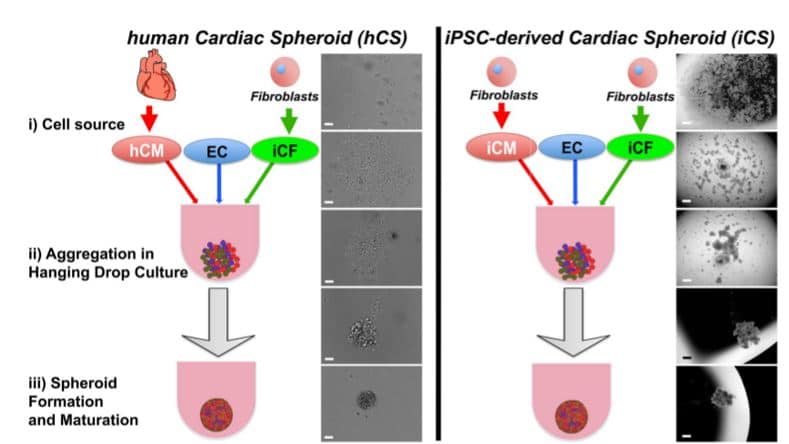
Figure 4. Spheroid formation in a hanging drop culture (Polonchuk et al., 2017)
Microfluidic devices
Finally, adding a microfluidic option to the previous technologies enables to reproduce in a more realistic way the physiology of cells by means of controlled flows (e.g. cell medium, drugs, etc). Also, the study of vascular models becomes easier with microfluidic systems, which could replace a part of animal experimentations (Suter-Dick et al., 2015). For instance, it has been reported in the literature that using this kind of device to test drugs known to induce cardiotoxicity (doxorubicin, verapamil and quinidine), the beating rate is decreased compared to 2D cell cultures (Bergström et al., 2015).
SmartHeart plates
4Dcell has developed an inovative solution to obtain reproducible cardiac organ-on-chips in an easy to use multiwell format (Organs in a well). These 3D structures are composed of soft materials that funnel the sedimenting cells towards a ring shape. As the cells attach to the bottom of the ring, they form a circular tissue around a central pillar. As the tissue matures and begins to contract, the beat rate is easily identifiable by the deformation of this central pillar. Furthermore, the mechanical properties of this pillar are very well characterized making it easy to determine the forces exerted by the cardiac tissue. To learn more about these structures, visit 4Dcell’s cardiac assay page.
> Impact of better preclinical models on drug screening
> Cardiotoxicity induced by drugs
Cardiotoxicity developed by drugs manifests mainly through ventricular arrhythmias, such as, Torsades de Pointes (TdP) by interfering with ion channel functions (Redfern et al., 2003).
To avoid the cardiotoxic effects of drugs on patients, safety pharmacology studies have been developed. For instance, the European Medicines Agency implemented regulamentation that protects participants during clinical trials (ICH S7A). More specifically, there is a norm to protect them, under a nonclinical evaluation, from ventricular repolarization and QT interval prolongation (ICH S7B). They are also under control with guidelines during clinical developments (ICH E14). Since 2013, to avoid and to detect TdP earlier and more confidently, comprehensive in vitro proarrythmia assay (CIPA) will likely update S7B and E14 guidances (Gintan et al., 2017).
In addition, CMs contractility capacities are also impacted by the negative effects that drugs can develop, such as, oncology drugs (Mellor et al., 2011). For that reason, the development of contractility assays is an urgent need since it can reduce the risk of drug cardiotoxicity.
> Cell technologies benefits
Cardiac repair
When heart failure occurs, it causes myocardial damages usually irreversible and difficult to treat (Mann, 1999). In this case, tissues obtained from tissue engineering can be an alternative to cardiac transplantations and overcome the issues to be a limited source depending of the donors. For instance, porous 3D alginate scaffolds enable aggregates formation where cardiac cells remain viable (Leor et al., 2000). Also, in a recent study iPSC-CMs from patients’ somatic tissues, where encapsulated in copolymer gel allowing to the LV chamber to be reduced, to the LV free wall to become thicker and the decrease of fibrosis (Wang et al., 2016) (Figure 4).
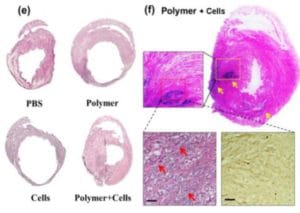
Figure 5.The effects of hydrogel when iPSC-CMs are encapsulated inside (Wang et al., 2016)
Heart function
Bioengineered cardiac tissues development has shown good opportunities to improve cardiac function. The major one is the vascularization of the cells because it enables to increase the viability of cells. In that way, cell sheets technology was developed. Also, in addition to the vascularization, which is improved, it decreases fibrosis and apoptosis. Studies have been performed to show that cell survival rate is improved compared to cell injection method (Sekine et al., 2011) leading to the improvement of the functions of cells.
Beating in a dish
One of the goals of innovative cardiac cells culture technologies is to observe a spontaneous contractility. In fact, in beating cardiomyocytes conditions improve the capacity of blood to circulate through the organ like in vivo (Sasaki et al., 2018). Using iPSC-CMs, the electrophysiologic phenotypes are closely comparable to the in vivo conditions which makes the beating rate similar (Karakikes et al., 2015).
> Perspectives
Among the recent evolution of the technology more studies need to be done. By improving in vitro cell models, we will obtain more representative assays for cardiotoxicity studies.
It has been found that kinase can lead to many diseases (specially cancers). That is the reason why, kinase inhibitor drugs have been developed. However, the main cause of failure of those drugs is the cardiotoxicity induced by kinase inhibitors (Noble et al., 2004). It is difficult to predict kinase inhibitors toxicity (Force et al., 2011) due to excitation-contraction impairs as reported by Peters et al.. Recently, it has been shown that by detecting cellular impedance it could improve the predictability of the kinase inhibitors toxicity by measuring beating rates, known, it needs to be further explored to have relevant results (Lamore et al., 2017). Impedance measurements are used in a regular basis for drug development (Peters et al., 2015).
In addition, to move forward, improvements in the protocol of hiPSC and cardiomyocytes need to be fixed to get a clear method of differentiation and maturation (Hirt et al., 2014).
Finally, another perspective could be the integration of cardiac tissues in vitro with structural properties combined to more functional blood vessels to enable the transport of drugs and detect easily the cardiotoxicity during drug development (Novakovic, 2014).
> Conclusion
To conclude, there is an urgent need to decrease drug withdrawal related to drug toxicity. To improve the measurement and the predictability of the effects of drugs on cardiac cells, new cell models associated to new technologies are being developed. 4Dcell works on advanced biotechnology to enable the improvement of preclinical models for drug screening, also including cardio-toxicity models.
> References
Bergström, G., Christoffersson, J., Schwanke, K., Zweigerdt, R., & Mandenius, C. F. (2015). Stem cell derived in vivo-like human cardiac bodies in a microfluidic device for toxicity testing by beating frequency imaging. Lab on a Chip, 15(15), 3242-3249.
Carter, S. B. (1967). Haptotactic islands – a method of confining single cells to study individual cell reactions and clone formation. Exp. Cell Res. 48, 189-193.
Dvir, T., Timko, B. P., Kohane, D. S., & Langer, R. (2011). Nanotechnological strategies for engineering complex tissues. Nature nanotechnology, 6(1), 13.
Eschenhagen, T., Bolli, R., Braun, T., Field, L. J., Fleischmann, B. K., Frisén, J., … & Marbán, E. (2017). Cardiomyocyte regeneration: a consensus statement. Circulation, 136(7), 680-686.
Force, T., & Kolaja, K. L. (2011). Cardiotoxicity of kinase inhibitors: the prediction and translation of preclinical models to clinical outcomes. Nature reviews Drug discovery, 10(2), 111.
Gintant, G., Fermini, B., Stockbridge, N., & Strauss, D. (2017). The evolving roles of human iPSC-derived cardiomyocytes in drug safety and discovery. Cell Stem Cell, 21(1), 14-17.
Hirt, M. N., Hansen, A., & Eschenhagen, T. (2014). Cardiac tissue engineering: state of the art. Circulation research, 114(2), 354-367.
Karakikes, I., Ameen, M., Termglinchan, V., & Wu, J. C. (2015). Human induced pluripotent stem cell–derived cardiomyocytes: insights into molecular, cellular, and functional phenotypes. Circulation research, 117(1), 80-88.noble
Kelm, J. M., Timmins, N. E., Brown, C. J., Fussenegger, M., & Nielsen, L. K. (2003). Method for generation of homogeneous multicellular tumor spheroids applicable to a wide variety of cell types. Biotechnology and bioengineering, 83(2), 173-180.
Lamore, S. D., Ahlberg, E., Boyer, S., Lamb, M. L., Hortigon-Vinagre, M. P., Rodriguez, V., … & Choy, A. L. (2017). Deconvoluting kinase inhibitor induced cardiotoxicity. Toxicological Sciences, 158(1), 213-226.
Langley, G., Austin, C. P., Balapure, A. K., Birnbaum, L. S., Bucher, J. R., Fentem, J., … & Lidbury, B. A. (2015). Lessons from toxicology: developing a 21st-century paradigm for medical research. Environmental health perspectives, 123(11), A268-A272.
Leor, J., Aboulafia-Etzion, S., Dar, A., Shapiro, L., Barbash, I. M., Battler, A., … & Cohen, S. (2000). Bioengineered cardiac grafts: a new approach to repair the infarcted myocardium?. Circulation, 102(suppl_3), Iii-56.
Li, D., Wu, J., Bai, Y., Zhao, X., & Liu, L. (2014). Isolation and culture of adult mouse cardiomyocytes for cell signaling and in vitro cardiac hypertrophy. JoVE (Journal of Visualized Experiments), (87), e51357.
Louch, W. E., Sheehan, K. A., & Wolska, B. M. (2011). Methods in cardiomyocyte isolation, culture, and gene transfer. Journal of molecular and cellular cardiology, 51(3), 288-298.
Mann, D. L. (1999). Mechanisms and models in heart failure: a combinatorial approach. Circulation, 100(9), 999-1008.
Masuda, S., & Shimizu, T. (2016). Three-dimensional cardiac tissue fabrication based on cell sheet technology. Advanced drug delivery reviews, 96, 103-109.
Mellor, K. M., Bell, J. R., Young, M. J., Ritchie, R. H., & Delbridge, L. M. (2011). Myocardial autophagy activation and suppressed survival signaling is associated with insulin resistance in fructose-fed mice. Journal of molecular and cellular cardiology, 50(6), 1035-1043.
Noble, M. E., Endicott, J. A., & Johnson, L. N. (2004). Protein kinase inhibitors: insights into drug design from structure. Science, 303(5665), 1800-1805.
Novakovic, G. V., Eschenhagen, T., & Mummery, C. (2014). Myocardial tissue engineering: in vitro models. Cold Spring Harbor perspectives in medicine, 4(3), a014076.
Peters, M. F., Lamore, S. D., Guo, L., Scott, C. W., & Kolaja, K. L. (2015). Human stem cell-derived cardiomyocytes in cellular impedance assays: bringing cardiotoxicity screening to the front line. Cardiovascular Toxicology, 15(2), 127-139.
Polonchuk, L., Chabria, M., Badi, L., Hoflack, J. C., Figtree, G., Davies, M. J., & Gentile, C. (2017). Cardiac spheroids as promising in vitro models to study the human heart microenvironment. Scientific reports, 7(1), 7005.
Redfern, W. S., Carlsson, L., Davis, A. S., Lynch, W. G., MacKenzie, I., Palethorpe, S., … & Camm, A. J. (2003). Relationships between preclinical cardiac electrophysiology, clinical QT interval prolongation and torsade de pointes for a broad range of drugs: evidence for a provisional safety margin in drug development. Cardiovascular research, 58(1), 32-45.
Ribeiro, A. J., Ang, Y. S., Fu, J. D., Rivas, R. N., Mohamed, T. M., Higgs, G. C., … & Pruitt, B. L. (2015). Contractility of single cardiomyocytes differentiated from pluripotent stem cells depends on physiological shape and substrate stiffness. Proceedings of the National Academy of Sciences, 112(41), 12705-12710.
Salick, M. R., Napiwocki, B. N., Sha, J., Knight, G. T., Chindhy, S. A., Kamp, T. J., … & Crone, W. C. (2014). Micropattern width dependent sarcomere development in human ESC-derived cardiomyocytes. Biomaterials, 35(15), 4454-4464.
Sasaki, D., Matsuura, K., Seta, H., Haraguchi, Y., Okano, T., & Shimizu, T. (2018). Contractile force measurement of human induced pluripotent stem cell-derived cardiac cell sheet-tissue. PloS one, 13(5), e0198026.
Sekine, H., Shimizu, T., Dobashi, I., Matsuura, K., Hagiwara, N., Takahashi, M., … & Okano, T. (2011). Cardiac cell sheet transplantation improves damaged heart function via superior cell survival in comparison with dissociated cell injection. Tissue engineering Part A, 17(23-24), 2973-2980.
Shimizu, T., Yamato, M., Isoi, Y., Akutsu, T., Setomaru, T., Abe, K., … & Okano, T. (2002). Fabrication of pulsatile cardiac tissue grafts using a novel 3-dimensional cell sheet manipulation technique and temperature-responsive cell culture surfaces. Circulation research, 90(3), e40-e48.yan
Snir, M., Kehat, I., Gepstein, A., Coleman, R., Itskovitz-Eldor, J., Livne, E., & Gepstein, L. (2003). Assessment of the ultrastructural and proliferative properties of human embryonic stem cell-derived cardiomyocytes. American Journal of Physiology-Heart and Circulatory Physiology, 285(6), H2355-H2363.
Soonpaa, M. H., & Field, L. J. (1998). Survey of studies examining mammalian cardiomyocyte DNA synthesis. Circulation research, 83(1), 15-26.
Stevens, J. L., & Baker, T. K. (2009). The future of drug safety testing: expanding the view and narrowing the focus. Drug discovery today, 14(3-4), 162-167.
Suter-Dick, L., Alves, P. M., Blaauboer, B. J., Bremm, K. D., Brito, C., Coecke, S., … & Jennings, P. (2015). Stem cell-derived systems in toxicology assessment. Stem cells and development, 24(11), 1284-1296.
Takahashi, K., & Yamanaka, S. (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. cell, 126(4), 663-676.
Tiburcy, M., Meyer, T., Soong, P. L., & Zimmermann, W. H. (2014). Collagen-based engineered heart muscle. In Cardiac Tissue Engineering (pp. 167-176). Humana Press, New York, NY.
Thomson, J. A., Itskovitz-Eldor, J., Shapiro, S. S., Waknitz, M. A., Swiergiel, J. J., Marshall, V. S., & Jones, J. M. (1998). Embryonic stem cell lines derived from human blastocysts. science, 282(5391), 1145-1147.
Wang, X., Chun, Y. W., Zhong, L., Chiusa, M., Balikov, D. A., Frist, A. Y., … & Sung, H. J. (2015). A temperature-sensitive, self-adhesive hydrogel to deliver iPSC-derived cardiomyocytes for heart repair. International journal of cardiology, 190, 177-180.
Weinberg, C. B., & Bell, E. (1986). A blood vessel model constructed from collagen and cultured vascular cells. Science, 231(4736), 397-400.
Wert, G. D., & Mummery, C. (2003). Human embryonic stem cells: research, ethics and policy. Human reproduction, 18(4), 672-682.
Yan, Y., Bejoy, J., Xia, J., Griffin, K., Guan, J., & Li, Y. (2019). Cell population balance of cardiovascular spheroids derived from human induced pluripotent stem cells. Scientific reports, 9(1), 1295.
Zimmermann, W. H., Schneiderbanger, K., Schubert, P., Didie, M., Munzel, F., Heubach, J. F., … & Eschenhagen, T. (2002). Tissue engineering of a differentiated cardiac muscle construct. Circulation research, 90(2), 223-230.
Zuppinger, C., Gibbons, G., Dutta-Passecker, P., Segiser, A., Most, H., & Suter, T. M. (2017). Characterization of cytoskeleton features and maturation status of cultured human iPSC-derived cardiomyocytes. European journal of histochemistry: EJH, 61(2).