> ORGAN IN WELL & HEPATOTOXICITY
New technologies for the prediction of hepatotoxicity during preclinical studies
> ORGAN IN WELL & HEPATOTOXICITY
New technologies for the prediction of hepatotoxicity during preclinical studies
> Introduction
Considered as the main reason to remove drugs from the market (Temple et al., 2002), drug induced liver injury (DILI) is a major concern of pharmaceuticals industries to decrease the hepatotoxicity of drugs before to arrive on the market. Consequences of hepatocyte damages usually affect inflammatory reactions and adaptive immune responses which causes the loss of the liver cells capacity to counteract the effect of the toxicity (Holt et al., 2006). For instance, necrosis, hepatitis, cholestasis or cirrhosis are outcomes of hepatotoxicity (Tujios et al., 2011). For those reasons, new cell models combined to new technologies are developed by 4Dcell to get a better cell characterization enabling a better prediction and quantification of DILI.
Two main elements are studied for hepatotoxicity: the cytochrome P 450 (CYP) activity and the bile canaliculi formation. P450 enzymes usually convert the drug into chemical compounds but if not, toxicity is developed (Park et al., 1995). Also, hepatotoxicity by drugs could inhibit the active transport of bile components like bile acids through canaliculi (Kostrubsky et al., 2003). So, by studying either the evolution of CYP activity or bile acid transport, relevant characteristics of hepatotoxicity can be obtained.
4Dcell develops in this sense new technologies where the 3Rs strategy (Replacement, Reduction, Refinement) is promoted decreasing the amount of animal studies and consequently the ethical issues due to the use of animal models (Langley et al., 2015).
This review aims to provide a state of the art of models and technologies developed to obtain a greater preclinical model to study hepatotoxicity.
> Cell models
> Primary cells
Primary Human Hepatocytes (PHH) are isolated from donors of liver material in accordance with the Declaration of Helsinki (1975) and requires a written consent. Considered as a gold standard for in vitro pharmacological studies, this cell model is often used for metabolism researches (Gómez-Lechón et al., 2014). Animal hepatocytes may be used to study metabolic pathways, but interspecies differences complicate the validation of a reaction from animals to humans. The most significant difference for drug metabolism is the CYP activities that are crucial making the extrapolation of results from animal to Human difficult (O’Brien et al., 2004).
However, PHH in culture rapidly lose their characteristics such as morphology, function or structure rapidly (Bell et al., 2015). Scarcity of this type of cells and their short lifespan make them less useful that they could be (Hart et al., 2010).
> Hepatoma cell lines
To counteract the drawbacks of PHH, namely, to be able to keep the cells for longer in vitro studies, hepatoma cell lines have been developed. They do not mimic the PHH structure and functions which are reduced, but they have an unlimited lifespan and are easily more available than primary cells (Donato et al., 2013).
HepG2
The HepG2 cell line was obtained from biopsies of liver tumor of a 15-year-old Caucasian male in Argentina in 1975. Although it doesn’t mimic the hepatocytes functions such as expression of CYP 450 enzymes which is highly reduced compared to human hepatocytes (Wilkening et al., 2003), it allows in vitro studies for clinical research due to the advantages discussed above. In addition, bile-canaliculi structures are formed and can be detected for better hepatotoxicity studies (Sormunen et al., 1993).
HepaRG
The HepaRG cell line is the most relevant model in hepatotoxicity studies due to its more efficient metabolic reactions compared to other cell lines.
Isolated from a human carcinoma, HepaRG has been the first cell line to be susceptible to hepatitis B virus (HBV) infection and cells are very close in their morphology of human hepatocytes after proliferation and differentiation (Gripon et al., 2002). In addition, it has been shown that P450-activities are efficient using HepaRG cells making them a more relevant model to study metabolism and hepatotoxicity (Aninat et al., 2005) even if it does not mimic the exact activity of PHH (Sison-Young et al., 2015). Useful to study DILI mechanism and to evaluate the risk to develop DILI in the preclinical study make HepaRG cells the best cell model discovered for drug testing (Tomida et al., 2015).
To overcome the actual limitations of hepatoma cell lines, development of new culture systems has been explored to improve the capacities of these models in a more efficient way.
> Cell culture systems
> 2D culture models
Hepatocyte monolayer
Monolayer cultures are used for human hepatocytes and allow to the cells coated on extracellular matrix (ECM) proteins to maintain their functions such as the CYP activities (Gómez-Lechón et al., 2014). However, monolayers cultures are used with PHH and the limitations of this models cited above (I.1.) make the use of this system difficult.
Sandwich culture
Hepatocytes cultivated in sandwich cultures are between two layers of gelled ECM proteins. Cells mimic more the metabolism of human cells and can be study for a longer time (Dunn et al., 1991) especially for DILI studies (Tuschl et al., 2009). Cellular repolarization has been observed using sandwich cultures hepatocytes (SCH) (Bi et al., 2006) which enables the identification of membrane transporter linked to bile canaliculi structures. This will be relevant to understand mechanisms of hepatotoxicity (Meng, 2010). Although SCH is a good model for hepatic disposition, it cannot be used as a model for P450 enzymes activities because it does not enable to obtain many of P450 enzymes (Kimoto et al., 2011).
> 3D culture models
Hydrogel
Hepatocytes can be suspended in scaffold-free systems where they are embedded in non-adhesive hydrogels (Koebe et al., 1994). Many types of hydrogels have been developed the last few decades such as MatrigelTM issued from a mouse sarcoma and based on an extracellular matrice (ECM) (Kleinman et al., 1986) or hydrogels based on collagen. However, difficulty to transfer nutrients or xenobiotics make the use of hydrogels limited (Godoy et al., 2013).
Spheroid
Hepatocellular spheroid formation can occur by different ways such as by agitation, hanging drop cell culture or culture on non-adherent surfaces (Poloznikov et al., 2018). Using spheroids long-term metabolic assays are more relevant (Ohkura et al., 2014) due to their ability to remain active for at least 21 days (Arakawa et al., 2017). In that way, cytochrome activity tests can be performed (Figure 1). Spheroids are often formed into hydrogels allowing the formation of bile canaliculi structures used for drug research (Malinen et al., 2012). Although spheroids have potential for drug screening, this model shows some limitations. In fact, developed to better mimic tissues it does not reflect yet the exact functions of hepatocytes. So many studies are needed to see how it expresses the real tissue like by studying gene expression (Lin et al., 2008).
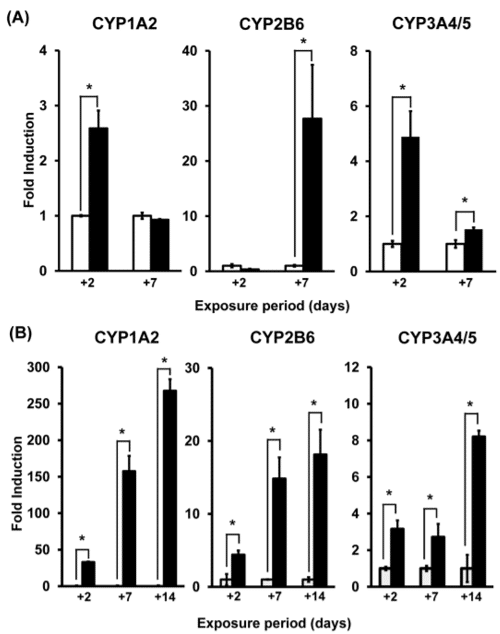
Figure 1. CYP activities in 2D (A) and in 3D (B)
In 3D cultures, CYP activities measured were maintained for a longer time than in 2D cultures.
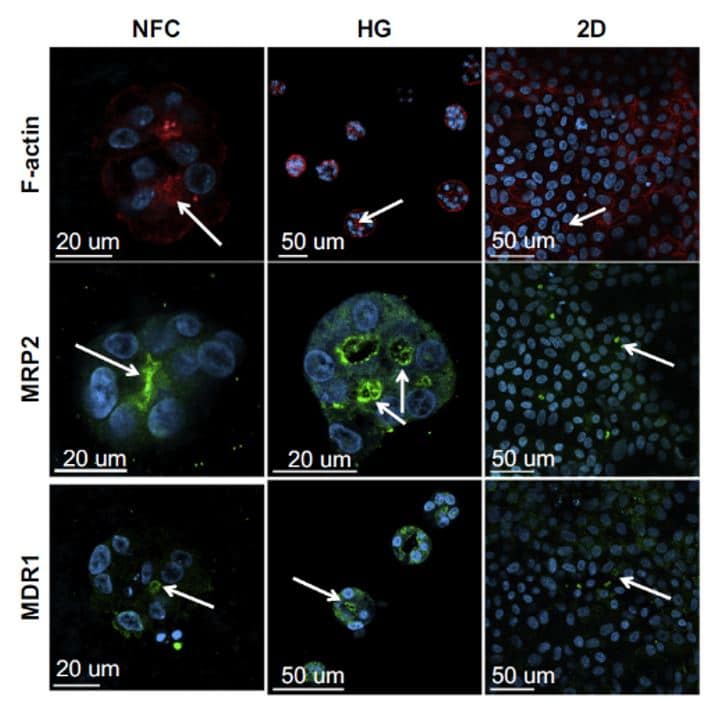
Figure 2. Spheroid formation in a soft substrate versus flat substrate
Accumulation of F-actin, MRP2 and MDR1, specific markers of the canalicular plasma membrane, have been detected in low-density HepaRGTM cultures using wood-derived nanofibrillar cellulose (NFC) and hyaluronan-gelatin (HG) hydrogels after spheroid formation compared to a 2D cell culture where the activity of MRP2 and MDR 1 is lower (Malinen et al., 2014).
Organ on-a-chip: Micropattern, microwell, microfluidic devices
To control the size of large amount of spheroid formed as described above is challenging. Because it limits the read-outs of the cell culture, micro-well technology has been developed to generate hepatocytes aggregates with a regular size. This organ on-a-chip device is used to enable a better expression of specific hepatocytes markers with specific hepatocytes dimensions between 90-120 micrometer diameter (Figure 3) (Gevaert et al., 2014).
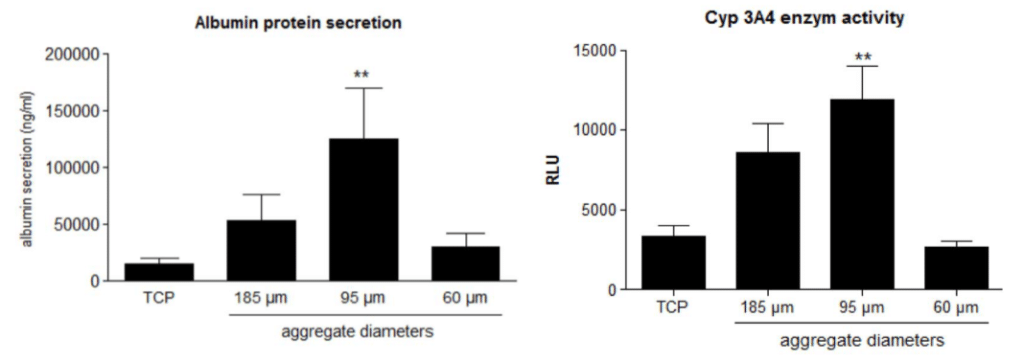
Figure 3. Albumin secretion and Cyp activity depending of aggregate diameters
Albumin protein secretion, Cyp 3A4 enzyme activity of cells have been measured using different diameters of aggregates. When the diameter corresponds to 95 µm, CYP activity and albumin secretion are higher compared to smaller or bigger diameters (Gevaert et al., 2014).
The idea to confine cells into delimited areas started 50 years ago in 1967 (Carter, 1967). However, it has been developed and used for 10 years with many kinds of cells including hepatocytes.
To ensure the lack of toxicity of a drug, bile canaliculi identification and quantification are major indicators, but it is difficult to identify bile canaliculi forming in between cells.
Using micropatterns, cells acquire the shape of the pattern where they are cultured. It enables a long-term confinement and optimizes cell-cell interaction for a better observation of bile canaliculi formation for a long time (Underhill et al., 2018) (4 weeks). Combining the technology of hydrogels with micropatterns enables to greater mimic the in vitro environment (Almany et al., 2005).
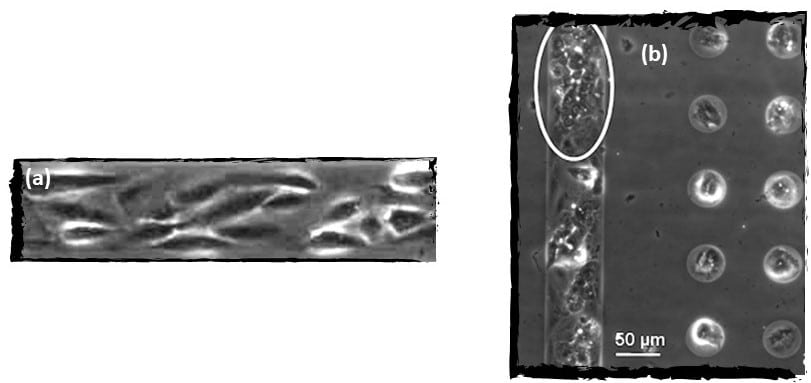
Figure 4. Organization of HepaRG cells after two days of culture (a) in line micropatterns in agarose substrate.
Observation of bile caniculae (bright) formed in between hepatocytes differentiated from HepaRG (b) (Mercey et al., 2010).
Microfluidic devices
Still focused on improving hepatocytes functions and viability in cell culture, microfluidic technology started to be used in the last few years. In this case human organs environment is reproduced through dynamic fluid flow and gradients (Esch, et al., 2015). Microphysiological systems (MPS) are based on this device to characterize the hepatocyte function when using it. Moreover, as living cells are able to transduce a biological response to a mechanical signal (Bao; Suresh, 2003), effect of shear stress can also be studied especially to measure the CYP450 activity and serum protein secretion in hepatocytes. For both characteristics measured in hepatocytes it improves the cells functions and phenotypes (Rashidi et al., 2016).
However, costs or scaling the data to approach as must as it is possible the real mechanism may be issues of this technique which need to be developed to have more relevant results (Chang et al., 2017).
> Preclinical models for drug screening
> Advantages of the new systems
The interests to exploit the potential of new cell models and new cell technologies are varied but mainly directed to improve cell environment as precisely as possible. Many applications areas can be covered with those technologies such as cancer research, organ physiology, immunology and so on. For hepatotoxicity, as we have developed previously, bile canaliculi formation and CYP activities are the most relevant data studied to prevent hepatotoxicity by drugs as earlier as possible. By combining new cell models to new technologies, preclinical assays will be more efficient to transduce in vivo mechanisms to in vitro. For instance, using HepaRGTM cells, cholestatic drug effects can be identified with the identification of bile canaliculi transporters (Figure 5) (Antherieu et al., 2013).
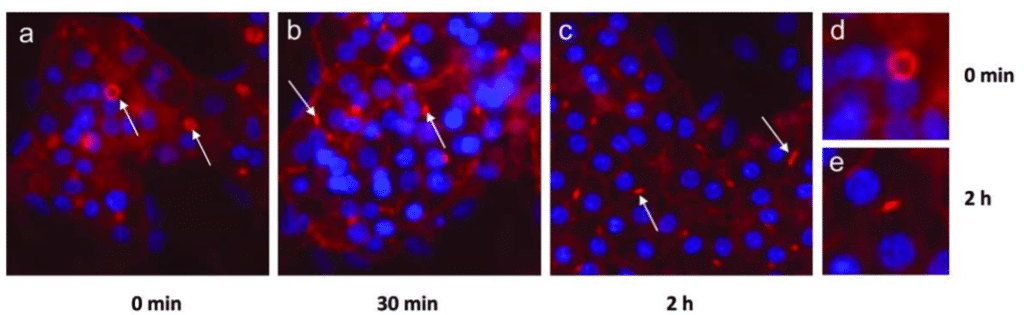
Figure 5. The effects of cholestatic drug: chlorpromazine (CPZ) on HepaRGTM cells.
After treatment with CPZ, known as an inductor of cholestasis, a decrease of bile canaliculi surface area is observed on HepaRGTM (Anthérieu et al., 2013).
> Solutions under development for better prediction
Predictive models have been developed for DILI and could be very efficient. From multiple sources, in silico can be obtained such as toxigenomic studies or chemical compounds (Chen et al., 2014). However, the lack of data is the main barrier to the efficacy of those models (He et al., 2019) because DILI mechanisms are difficult to identify and are diverse (Mulliner et al., 2016).
Biomarkers could also help to detect and to predict DILI. For instance, enzymes such as alanine aminotransferase (ALT) or Aspartate aminotransferase (AST) allow the detection of DILI after their quantification (Ozer et al., 2008). In addition, it has been shown that detections of miRNAs levels in the plasma could be used (Wang et al., 2009).
By detecting specific genetic predisposing factors to develop liver injury, it could improve predictability for adverse drug reactions (Pirmohamed, 2012). Improving genomic studies will lead to an improvement of the personalized medicine.
> Conclusion
To conclude, drug toxicity is one of the consequences of drug metabolism and that is the reason why, metabolic activity studies are crucial. To improve the measurement and the predictability of the drug effects on hepatocytes, new cell models associated to new technologies are developed. 4Dcell works with advancing science and technologies to enable the improvement of preclinical model for drug screening.
> References
Almany, L., & Seliktar, D. (2005). Biosynthetic hydrogel scaffolds made from fibrinogen and polyethylene glycol for 3D cell cultures. Biomaterials, 26(15), 2467-2477.
Aninat, C., Piton, A., Glaise, D., Le Charpentier, T., Langouët, S., Morel, F., … & Guillouzo, A. (2006). Expression of cytochromes P450, conjugating enzymes and nuclear receptors in human hepatoma HepaRG cells. Drug Metabolism and Disposition, 34(1), 75-83.
Anthérieu, S., Azzi, P. B. E., Dumont, J., Abdel‐Razzak, Z., Guguen‐Guillouzo, C., Fromenty, B., … & Guillouzo, A. (2013). Oxidative stress plays a major role in chlorpromazine‐induced cholestasis in human HepaRG cells. Hepatology, 57(4), 1518-1529.
Arakawa, H., Kamioka, H., Jomura, T. et al. (2017). Preliminary Evaluation of Three-Dimensional Primary Human Hepatocyte Culture System for Assay of Drug-Metabolizing Enzyme-Inducing Potential. Biological & Pharmaceutical Bulletin 40, 967–974. doi:10.1248/bpb.b16-00885
Bao, G., & Suresh, S. (2003). Cell and molecular mechanics of biological materials. Nature materials, 2(11), 715.
Bell, C. C., Hendriks, D. F., Moro, S. M., Ellis, E., Walsh, J., Renblom, A., … & Sison-Young, R. L. (2016). Characterization of primary human hepatocyte spheroids as a model system for drug-induced liver injury, liver function and disease. Scientific reports, 6, 25187.
Bi, Y.-a., Kazolias, D., and Duignan, D. B. (2006) Use of cryopreserved human hepatocytes in sandwich culture to measure hepatobiliary transport. Drug Metab. Dispos. 34, 1658−1665.
Carter, S. B. (1967). Haptotactic islands – a method of confining single cells to study individual cell reactions and clone formation. Exp. Cell Res. 48, 189-193.
Chang, S. Y., Voellinger, J. L., Van Ness, K. P., Chapron, B., Shaffer, R. M., Neumann, T., … & Eaton, D. L. (2017). Characterization of rat or human hepatocytes cultured in microphysiological systems (MPS) to identify hepatotoxicity. Toxicology in Vitro, 40, 170-183.
Chen, M., Bisgin, H., Tong, L., Hong, H., Fang, H., Borlak, J., & Tong, W. (2014). Toward predictive models for drug-induced liver injury in humans: are we there yet?. Biomarkers in medicine, 8(2), 201-213.
Teresa Donato, M., Jover, R., & Jose Gomez-Lechon, M. (2013). Hepatic cell lines for drug hepatotoxicity testing: limitations and strategies to upgrade their metabolic competence by gene engineering. Current drug metabolism, 14(9), 946-968.
Dunn, J. C., Tompkins, R. G. and Yarmush, M. L. (1991). Long-term in vitro function of adult hepatocytes in a collagen sandwich configuration. Biotechnol. Prog. 7(3):237–245.
Esch, E.W., A. Bahinski, and D. Huh, Organs-on-chips at the frontiers of drug discovery. Nat Rev Drug Discov, 2015. 14(4): p. 248-60.
Gevaert, E., Dolle, L., Billiet, T., Dubruel, P., van Grunsven, L., van Apeldoorn, A., & Cornelissen, R. (2014). High throughput micro-well generation of hepatocyte micro-aggregates for tissue engineering. PLoS One, 9(8), e105171.
Godoy, P., Hewitt, N. J., Albrecht, U., Andersen, M. E., Ansari, N., Bhattacharya, S., … & Braeuning, A. (2013). Recent advances in 2D and 3D in vitro systems using primary hepatocytes, alternative hepatocyte sources and non-parenchymal liver cells and their use in investigating mechanisms of hepatotoxicity, cell signaling and ADME. Archives of toxicology, 87(8), 1315-1530.
Gómez-Lechón, M. J., Tolosa, L., Conde, I., & Donato, M. T. (2014). Competency of different cell models to predict human hepatotoxic drugs. Expert opinion on drug metabolism & toxicology, 10(11), 1553-1568.
Gripon P, Rumin S, Urban S, Le Seyec J, Glaise D, Cannie I, Guyomard C, Lucas J, Trepo C, and Guguen-Guillouzo C (2002) Infection of a human hepatoma cell line by hepatitis B virus.
Proc Natl Acad Sci USA 99:15655–15660.
Hart, S. N., Li, Y., Nakamoto, K., Subileau, E. A., Steen, D., & Zhong, X. B. (2010). A comparison of whole genome gene expression profiles of HepaRG cells and HepG2 cells to primary human hepatocytes and human liver tissues. Drug Metabolism and Disposition, 38(6), 988-994.
Holt, M. P., & Ju, C. (2006). Mechanisms of drug-induced liver injury. The AAPS journal, 8(1), E48-E54.
Kimoto, E., Walsky, R., Zhang, H., Bi, Y. A., Whalen, K. M., Yang, Y. S., … & El-Kattan, A. F. (2012). Differential modulation of cytochrome P450 activity and the effect of 1-aminobenzotriazole on hepatic transport in sandwich-cultured human hepatocytes. Drug Metabolism and Disposition, 40(2), 407-411.
Kleinman HK, McGarvey ML, Hassell JR, Star VL, Cannon FB, Laurie GW, Martin GR (1986) Basement membrane complexes with biological activity. Biochemistry 25:312–318.
Koebe HG, Pahernik S, Eyer P, Schildberg FW (1994) Collagen gel immobilization: a useful cell culture technique for long-term metabolic studies on human hepatocytes. Xenobiotica 24:95–107.
Kostrubsky, V. E., Strom, S. C., Hanson, J., Urda, E., Rose, K., Burliegh, J., … & Sahi, J. (2003). Evaluation of hepatotoxic potential of drugs by inhibition of bile-acid transport in cultured primary human hepatocytes and intact rats. Toxicological Sciences, 76(1), 220-228.
Langley, G., Austin, C. P., Balapure, A. K., Birnbaum, L. S., Bucher, J. R., Fentem, J., … & Lidbury, B. A. (2015). Lessons from toxicology: developing a 21st-century paradigm for medical research. Environmental health perspectives, 123(11), A268-A272.
Lin, R. Z., & Chang, H. Y. (2008). Recent advances in three‐dimensional multicellular spheroid culture for biomedical research. Biotechnology Journal: Healthcare Nutrition Technology, 3(9‐10), 1172-1184.
Malinen, M. M., Palokangas, H., Yliperttula, M., & Urtti, A. (2012). Peptide nanofiber hydrogel induces formation of bile canaliculi structures in three-dimensional hepatic cell culture. Tissue Engineering Part A, 18(23-24), 2418-2425.
Meng Q. Three-dimensional culture of hepatocytes for prediction of druginduced hepatotoxicity. Expert Opin Metab Toxicol 2010;6:733-46.
Mercey, E., Obeïd, P., Glaise, D., Calvo-Muñoz, M. L., Guguen-Guillouzo, C., & Fouqué, B. (2010). The application of 3D micropatterning of agarose substrate for cell culture and in situ comet assays. Biomaterials, 31(12), 3156-3165.
Mulliner, D.; Schmidt, F.; Stolte, M.; Spirkl, H.P.; Czich, A.; Amberg, A. Computational Models for Human and
Animal Hepatotoxicity with a Global Application Scope. Chem. Res. Toxicol. 2016, 29, 757–767.
O’Brien, P. J., Chan, K., Silber, P. M. (2004). Human and animal hepatocytes in vitro with extrapolation in vivo. Chem. Biol. Interact. 150:97–114.
Ohkura T, Ohta K, Nagao T, Kusumoto K, Koeda A, Ueda T, Jomura T, Ikeya T, Ozeki E, Wada K, Naitoh K, Inoue Y, Takahashi N, Iwai H, Arakawa H, Ogihara T. Evaluation of human hepatocytes cultured by three-dimensional spheroid systems for drug metabolism. Drug Metab. Pharmacokinet., 29, 373–378 (2014)
Ozer, J., Ratner, M., Shaw, M., Bailey, W., & Schomaker, S. (2008). The current state of serum biomarkers of hepatotoxicity. Toxicology, 245(3), 194-205.
Park, B. K., Pirmohamed, M., & Kitteringham, N. R. (1995). The role of cytochrome P450 enzymes in hepatic and extrahepatic human drug toxicity. Pharmacology & therapeutics, 68(3), 385-424.
Pirmohamed, M. (2012). Genetics and the potential for predictive tests in adverse drug reactions. In Adverse Cutaneous Drug Eruptions (Vol. 97, pp. 18-31). Karger Publishers.
Poloznikov, A., Gazaryan, I., Shkurnikov, M., Nikulin, S., Drapkina, O., Baranova, A., & Tonevitsky, A. (2018). In vitro and in silico liver models: Current trends, challenges and opportunities. ALTEX-Alternatives to animal experimentation, 35(3), 397-412.
Rashidi, H., Alhaque, S., Szkolnicka, D., Flint, O., & Hay, D. C. (2016). Fluid shear stress modulation of hepatocyte-like cell function. Archives of toxicology, 90(7), 1757-1761.
Rowley JA, Madlambayan G, Mooney DJ (1999) Alginate hydrogels as synthetic extracellular matrix materials. Biomaterials 20:45–53.
Sormunen R, Eskelinen S, Lehto VP (1993) Bile canaliculus formation in cultured HEPG2 cells. Lab Invest 68:652–662.
Sison-Young, R. L. C., Mitsa, D., Jenkins, R. E. et al. (2015). Comparative Proteomic Characterization of 4 Human Liver-Derived Single Cell Culture Models Reveals Significant Variation in the Capacity for Drug Disposition, Bioactivation, and Detoxication. Toxicological Sciences : An Official Journal of the Society of Toxicology 147, 412–24.
Temple, R. J., & Himmel, M. H. (2002). Safety of newly approved drugs: implications for prescribing. Jama, 287(17), 2273-2275.
Tujios, S., & Fontana, R. J. (2011). Mechanisms of drug-induced liver injury: from bedside to bench. Nature reviews Gastroenterology & hepatology, 8(4), 202.
Tuschl, G., Hrach, J., Walter, Y., Hewitt, P. G., & Mueller, S. O. (2009). Serum-free collagen sandwich cultures of adult rat hepatocytes maintain liver-like properties long term: A valuable model for in vitro toxicity and drug–drug interaction studies. Chemico-biological interactions, 181(1), 124-137.
Tomida, T., Okamura, H., Satsukawa, M., Yokoi, T., & Konno, Y. (2015). Multiparametric assay using HepaRG cells for predicting drug-induced liver injury. Toxicology letters, 236(1), 16-24.
Underhill GH and Khetani SR (2018) Advances in engineered human liver platforms for drug metabolism studies. Drug Metab Dispos 46:1626–1637.
Wang, K., Zhang, S., Marzolf, B., Troisch, P., Brightman, A., Hu, Z., … & Galas, D. J. (2009). Circulating microRNAs, potential biomarkers for drug-induced liver injury. Proceedings of the National Academy of Sciences, 106(11), 4402-4407.
Wilkening, S., Stahl, F. and Bader, A. (2003). Comparison of primary human hepatocytes and hepatoma cell line Hepg2 with regard to their biotransformation properties. Drug Metabolism and Disposition: The Biological Fate of Chemicals 31, 1035–42.