Wound healing assay
How to measure wound closure over time using image analysis?
How to measure wound closure over time using image analysis?
Wound healing, % wound closure, wound closing rate, cell migration, area occupied by cells, image object detection
The wound healing assay is traditionally performed by scratching a cell monolayer to form a cell-free region (wound) and monitor cell migration into the wound. Although easy to set-up and inexpensive, this method creates inconsistent wounds with complicated geometries, which make the images difficult to analyze.
The method calculates the area occupied by the cells on the image. As cells migrate over time, the area occupied by the cells increases and the wound region becomes smaller until it is fully covered by cells. The method uses CellProfiler, a free, python-based open source software for biological images analysis. Note that it is also possible to use other softwares or to write a program from scratch to perform the analysis.
In this example, a wound healing assay was set-up on dynamic micropatterns. Mesenchymal stem cells were cultured on a 4DCELL 18 mm dynamic micropatterned glass coverslips, not coated with any ECM protein.
The micropatterned coverslips consisted of a rectangular shape of width 500 um coated with the anti-adhesive agent. 60’000 cells were seeded on the coverslip.
After 24 hours of incubation, the cells were homogeneously patterned in the regions surrounding the wound.
To activate migration into the wound, a high dose (50 uM) of the triggering reagent was added to render the wound region adhesive.
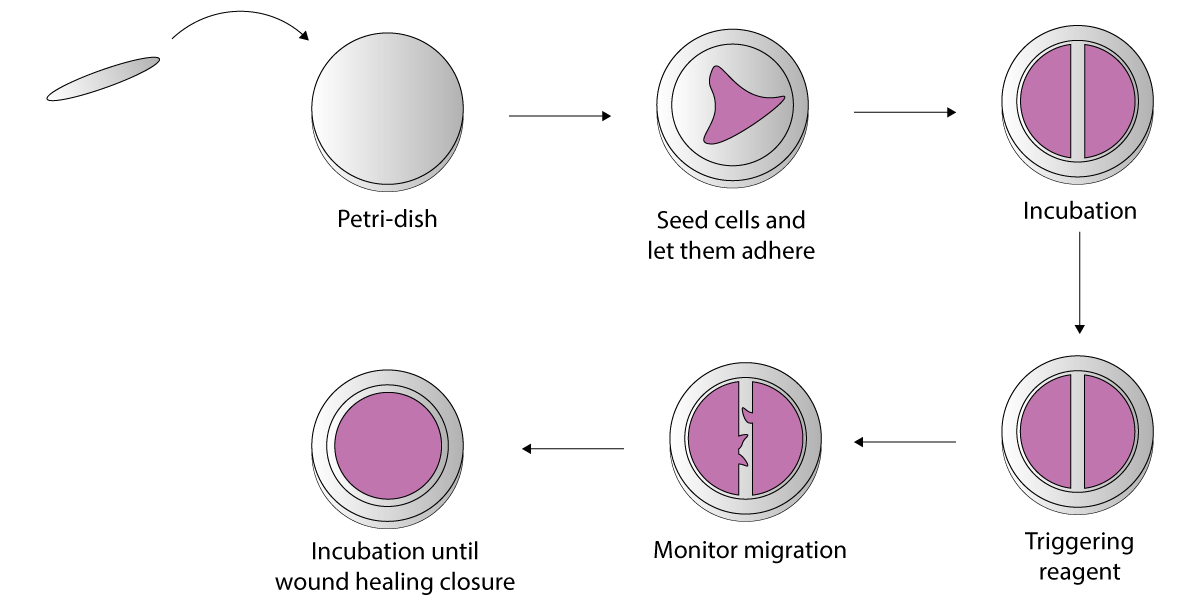
Setting up the wound healing assay using a 4DCELL dynamic micropatterned coverslip.
The rate of cell migration was quantified by measuring the area occupied by cells over time.
First, the background was subtracted from the image using the imageJ built-in ‘’rolling ball’’ algorithm. This allowed to enhance the contrast between cells and background and to correct for uneven illumination.
The image was converted to a grayscale image using the ‘ColorToGray’ CellProfiler module. The ‘Smooth’ module was then applied to the grayscale image to remove artifacts. Next, the ‘IdentifyPrimaryObjects’ and ‘MeasureImageAreaOccupied’ modules were successively applied. The first classifies features on the image into two two classes: object and background, where the cells correspond to objects. The latter measures the area taken by the previously calculated object on the image.
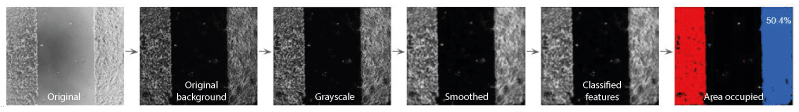
Sequence of modules used in the CellProfiler pipeline for measuring the area occupied by cells on the image
The area occupied by the cells on the original image is 50%.
Images of the substrate were acquired at three timepoints: immediately after triggering cell migration with the reagent, 8 hours after and 24 hours after. A non-treated negative control is also provided. The area occupied by cells was calculated using CellProfiler. On the treated sample, migration into the wound steadily increases and the wound is almost fully closed after 24 hours (98.6% closure). On the control sample the rate of wound closure is less. Partial wound closure on the non-treated sample after 24 hours is likely due to cell proliferation because the regions surrounding the wound are too confluent.

Setting up the wound healing assay using a 4DCELL dynamic micropatterned coverslip.
This simple example illustrates how it is possible to generate and analyze data using the 4DCELL dynamic micropatterned coverslips. Object detection by image segmentation is a powerful method to quantify the rate of wound closure on the 4DCELL wound healing assay.