> CELL MIGRATION DIRECTED BY MICROPATTERNED PROTEIN SUBSTRATES
These experiments illustrate the importance and concern of the 2D surface while doing cell culture on a dish. Cells sense and react to their environment, geometrically and chemically; they change their internal organization and adapt their external shape.
> Controlled 2D geometry
> Compatible with high resolution imaging and immunofluorescence
> Single cell analysis

HeLa cells plated on line micropatterns of 100 to 10 µm width, 4DCell Lab (2018).
PRINCIPLE AND BENEFITS
Nowadays micro-engineering techniques help to mimic the cell microenvironment by manipulating the cell culture conditions. Micropatterns can provide a dynamic, complex and micrometer-scale habitat as well as a good observation of the cell migration induced by different protein substrate shapes.
EXPERIMENTAL SETUP
This is a standard protocol for the micropatterns fabrication and can be adapted depending on the protein substrate and cell type.
> Material needed
Seeding substrate: Sterile micropatterned glass slides
Protein coating: fibronectin 50 µg/mL
Type of pattern: Triangles, squares and dots
> Protocol
Protein coating
On a parafilm, pipette 100 µL of fibronectin
Gently place the micropatterned side of the slide on the drop
Incubate 1 hour
Gently rinse the slide with water and then PBS
Cell seeding
Purified cells should be seeded onto micropatterns within 1 to 3 days after the purification step had been finished.
Wash the purified cells 3 times with PBS
Seed the cells at a density of 30 000 cells/cm² for confluent cell attachment
You can also refer to the dedicated User Guide.
RESULTS
Here you can find results from three different experiments using micropatterned protein substrates while observing and analyzing cell migration.
- Graphene Oxide micropatterns study
- Dynamic adhesion through a photo-activated surface
- Motion via asymmetric ratchet micropatterns
1. Graphene oxide micropatterns study
This experiment shows the relevance of the micropattern shape on the cell migration. The coating substrate made via meniscus-draging deposition (MDD) is Graphene oxide (GO), a hydrophilic carbon-based molecule, and the cells are L-929 fibroblasts.
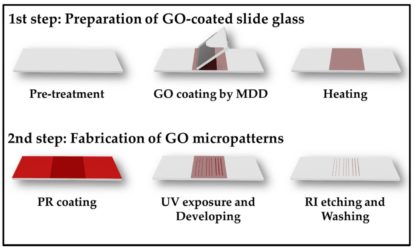
Figure 1: Preparation of the micropatterns in two main steps
(MDD: meniscus-draging deposition; PR : positive photoresists; UV: ultra-violet; RI: reactive-ion)
Observation of the cell migration for 12 hours
L-929 fibroblasts move rather on patterned area than unpatterned one
On square micropatterns, cells move from left to right only
On triangle micropatterns, cells move first from left to right then from right to left (this is due to the fact that the triangle has an asymmetric shape and induces a bidirectional path)
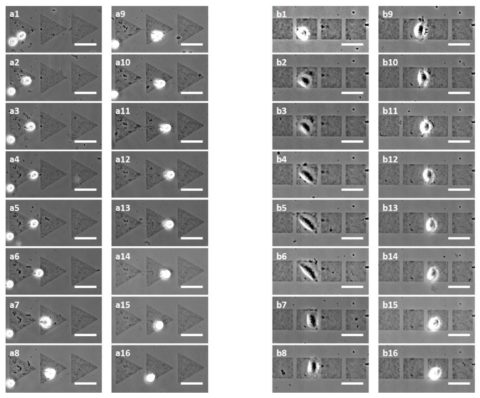
Figure 2: Time-lapse pictures of the L-929 fibroblasts motion for 12 hours
Quantitative analysis of the cell migration is illustrated on figure 3.
Trajectories are different on both micropatterns: on triangle ones, movement is oblique whereas on square ones, it is not
On triangle cells, speed and distance of the cells are higher than on square ones
Morphology changes due to the micropattern shape
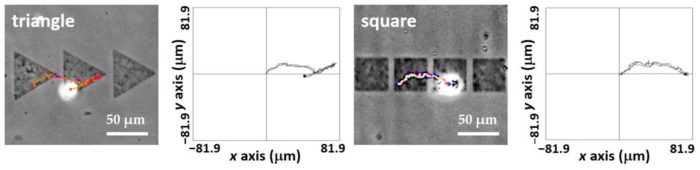
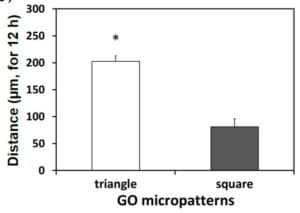
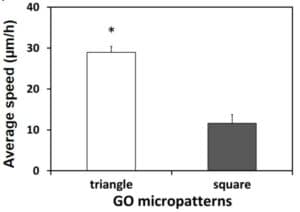
Figure 3: Representation of the cell trajectories on both micropattern shapes
These results demonstrate that the shape of the micropatterns has a significant impact on the cell speed, trajectory and motion. In addition, the triangle shape is more suitable to induce cell migration without any chemical influence than the square one.
2. Dynamic adhesion through a photo-activated surface
This experiment investigates the link between the cell attachment and the substrate with a spatiotemporal control of the anti-adhesive molecule.
The glass coverslip was modified prior patterning with NPE-TCSP having a photocleavable 2-nitrobenzyl group then incubated with BSA (see figure 4 for details).
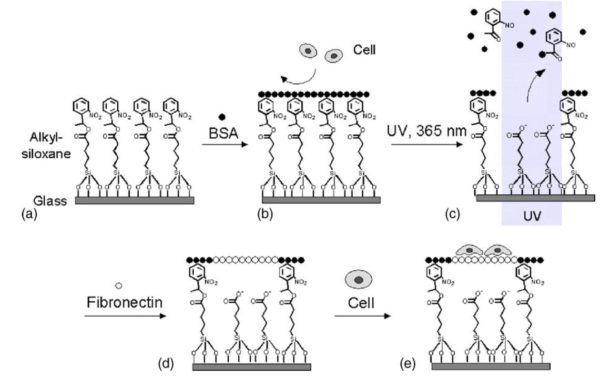
Figure 4: Representation of the micropatterning technique
Observation of cell migration for 49 hours
HEK293 cells are plated onto a fibronectin circular region and cultured for 28 hours
A second round area attached to the first one is illuminated by UV (no fibronectin incubation this time)
Cells migrate and proliferate towards this new region (see figure 5).
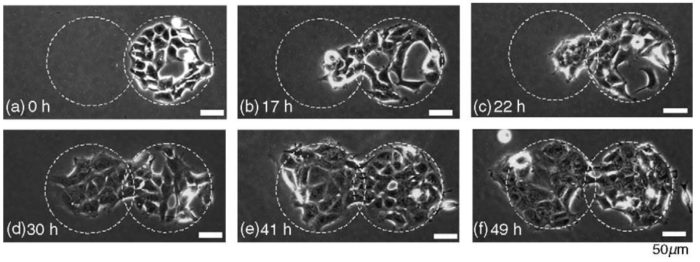
Figure 5: Time-lapse images of the HEK293 motion during 49 hours
These results demonstrate the impact of the anti-adhesive substrate on the cell adhesion.
3. Motion via asymmetric ratchet micropatterns
This experiment uses micropatterned agarose stamps and analyzes the cell motion directed by micropatterned ratchets.
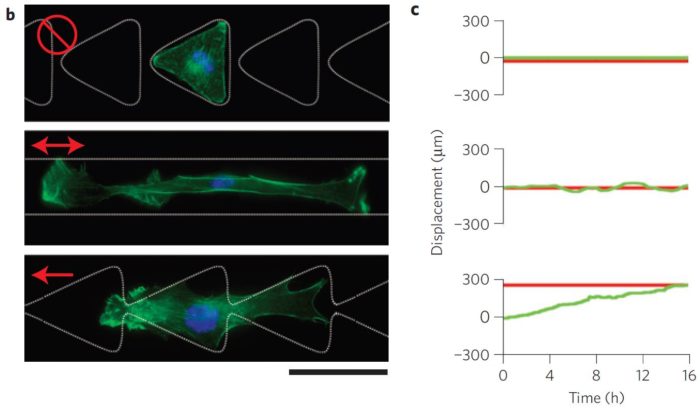
Figure 6: Illustration of the experiment
Cells on separate triangle micropatterns do not move at all
Cells on a straight line move in a horizontal way
Cells on linked triangle move in a predefined direction
Zooming into the interconnected ratchet micropatterns, a deeper analysis at the single cell level:
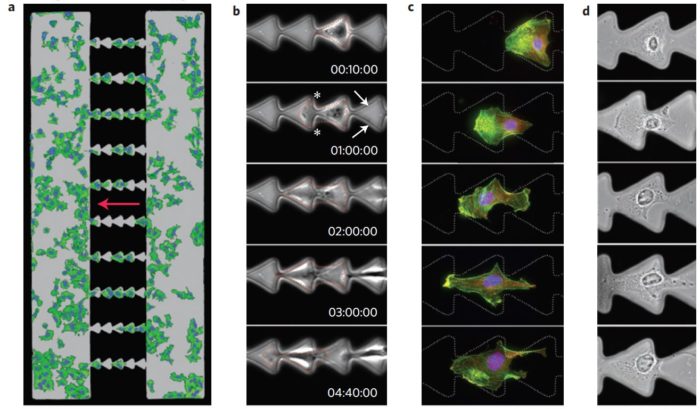
Figure 7: Cell migration induced by interconnected triangular shapes
These results demonstrate that the design of the micropatterned substrate of the adhesive molecule determine the direction of the cell migration.
CONCLUSION
Each of the above experiments reveal a clear influence of the micropatterning techniques on the cell culture on a dish. This 2D substrate modification has a crucial impact on the cell behavior and migration which is confirmed clearly with the experiment 2. We infer from the experiment 1 and 3 that triangular patterns fit more the cell migration study and analysis compared to square ones, for instance.
REFERENCES
Cell Migration According to Shape of Graphene Oxide Micropatterns
Sung Eun Kim, Min Sung Kim, Yong Cheol Shin, Seong Un Eom, Jong Ho Lee, Dong-Myeong Shin, Suck Won Hong, Bongju Kim, Jong-Chul Park, Bo Sung Shin, Dohyung Lim,and Dong-Wook Han, 2016.
Spatiotemporal control of cell adhesion on a self-assembled monolayer having a photocleavable protecting group
Jun Nakanishi, Yukiko Kikuchi, Tohru Takarada, Hidekazu Nakayama, Kazuo Yamaguchi and Mizuo Maeda, 2006.
Directing cell motions on micropatterned ratchets
Goher Mahmud, Christopher J. Campbell, Kyle J. M. Bishop, Yulia A. Komarova, Oleg Chaga, Siowling Soh, Sabil Huda, Kristiana Kandere-Grzybowska and Bartosz A. Grzybowski, 2009.
